The proposed super-resolution multiplexed line scan microscopy system realizes photon reassignment (PRA) microscopy technique for imaging fluorophore-labeled specimens. Unlike existing PRA techniques with a restricted two-fold resolution enhancement, the present system surpasses diffraction Abbe limit by a factor m=〖(σ_D/σ_E)〗^2+1>2, because standard deviations of an excitation line spread function (LSF) σE and of a detection point spread function (PSF) σD are substantially different: σE < σD. This difference is induced by combination of the focal line scanning and tailoring illumination LSF with the aid of pupil filters. The minimum detected spot width is σ_R=σ_D/√m. Further resolution m-fold improvement may be reached by deconvolution of the resulting PSF.
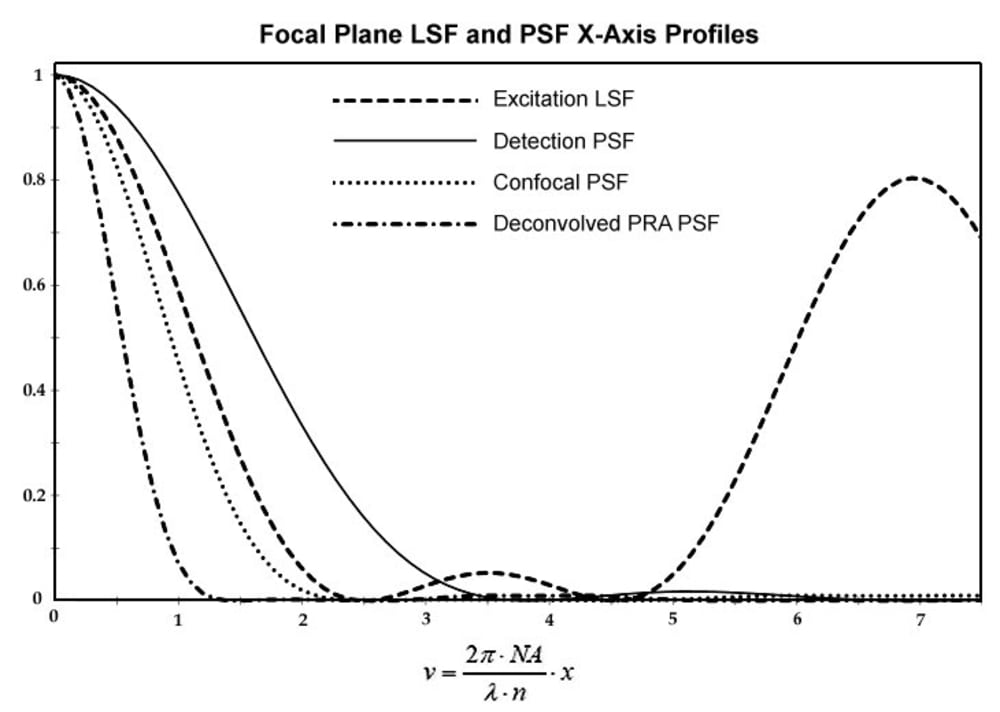
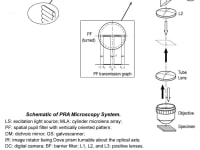
The system depicted in FIG. 1 provides scanning the specimen S with a plurality of spatially modulated with a pupil filter PF focal lines of excitation light in at least two distinct scan directions and detection of emission light from the specimen with high frame rate digital camera DC synchronized with the scanner GS. Each scan step is followed by the camera exposure, acquiring a partial confocal frame. Resulting image with m-fold enhanced isotropic lateral super-resolution is generated by joint digital postprocessing the partial images acquired with the digital camera in the process of scanning the specimen ROI. The approach allows imaging signal-to-noise ratio (SNR) better than one reachable in line scan confocal microscopy. The depicted exemplary PF, comprising two identical narrow light blocking strips symmetrically deposited along the pupil diameter, provides LSF with an isolated narrow central lobe and low intensity adjacent side lobes. FIG. 2 presents profiles of an excitation LSF, provided with the PF having, e.g., t1 = 0.8 and t2 = 0.13, of a pixel detection PSF, of a confocal PSF, i.e. joint reassignment PSF prior deconvolution, and deconvolved one. The Stokes shift is neglected. The microscopy system provides factor m = 3.3, corresponding to resolution δ = 66 nm, for NA = 1.4 and excitation λD = 500 nm. Application of a fluorophore with substantial Stokes shift, e.g., eFluor 506, results in δ < 50 nm. Imaging rate of the microscopy system may be sufficiently sped up by application of a refocusing MLA and a synchronized rescan mirror.
The proposed microscopy system is seen as a cost effective instrument, attractive and affordable for 2D and 3D in-vivo visualization of biological specimens at virus-scaled super-resolution, high SNR, and wide field of view, yielding a widely applicable tool for biomedical, forensic, and potential clinical applications.
The system may be manufactured of purchased off-the-shelf optomechanical components and 3D printed enclosures. Cost of all breadboard prototype components is below $6000, so the product is to be an order of value less expensive than STED microscopes having comparable resolution, whose prices start at $500,000. Therefore, it has a great market potential, because in the United States only, there are approximately 5,300 colleges and universities, whose research labs would be interested in the product.
Like this entry?
-
About the Entrant
- Name:Peter Vokhmin
- Type of entry:individual
- Software used for this entry:PTC Mathcad, ZEMAX
- Patent status:pending