Abstract:
Radiofrequency ablation (RFA) is a medical procedure used for treatment of cardiac disorders such as arrhythmia or destruction of cancerous tumors in lung, kidney, breast, liver or any other dysfunctional tissue diseases. RFA is also used for treatment of varicose veins, severe chronic pain as well as many other medical uses.
RFA Side Effects and Complications:
Although radiofrequency ablation is a relatively safe procedure, serious complication and side effects can occur including tissue charring and blood coagulation, blood clot and stroke, cardiac perforation, pulmonary vein stenosis, etc.
Solution:
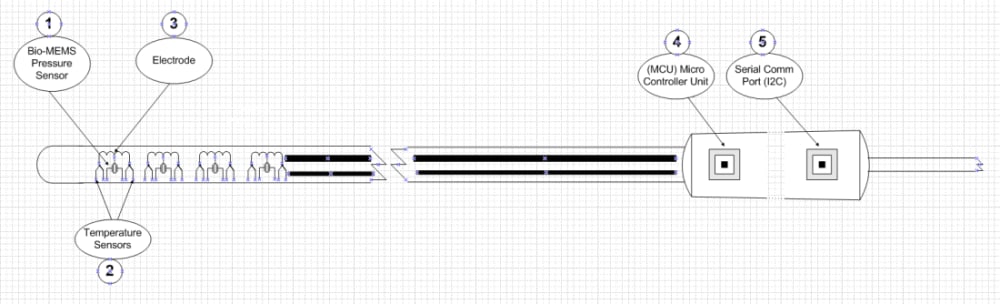
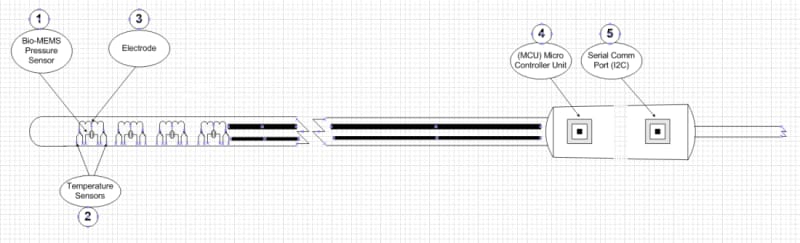
The conceptual design of the “Advanced Multi-Electrodes RFA Catheter” is based on a multi-point electrode design which monitors temperature and tissue contact quality. Electrode’s temperature is measured at both end points, and tissue contact pressure is monitored at the center of the ablating electrode using a Bio-MEMS ultra miniature pressure sensor embedded on the opposite side of the electrode. These critical measurements are the key to ensure tissue contact quality, and lesion transmurality. The physician is immediately able to validate electrode contact quality without additional RF. The surgical procedure is now significantly safer and consumes less time and RF energy.
Figure 1 depicts the conceptual design of this catheter including the Bio-MEMS ultra miniature pressure transducer (1) for monitoring the electrode’s mid-point force verifying tissue contact quality, temperature sensors (2) for precise monitoring of each electrode’s temperature, ablating electrode (3), micro controller (4) for data handling and processing and I2C data communication interface (5) to the RF generator.
Benefits:
+ Reduces ablation time and required RF energy improving patient safety factors
+ Provides precise feedback for electrode tissue contact quality in real time
+ Reduces electrode placement errors due to reliance on impedance-only feedback
+ Precise tissue temperature monitoring with two temperature sensors per electrode
+ Communicates with RF generator through I2C interface for vital information updates
+ Catheter’s manufacturer specifications are stored in a Flash ROM including catheter type, serial number, calibration data, etc. therefore reducing human error factors.
Like this entry?
-
About the Entrant
- Name:Michael Nasab
- Type of entry:individual
- Software used for this entry:MS Word, Power Point
- Patent status:pending