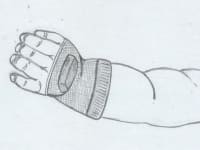
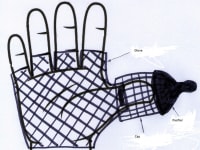
A safe, wireless, wearable device applicable to the finger or toe or hand palm in order to reduce the risk of the Sudden Infant Death syndrome. This device may be inside a fabric finger cap, perhaps with a pacifier, on a glove. It measures blood oxygen saturation and heart rate via pulse oximetry. When any of these parameters falls, an electric stimulation causes the child’s reaction. This reaction causes the child to save himself.
Actually, the FDA has not approved any medical device to reduce the risk of SIDS. A medical device must join two conditions to be approved. My device joins both.
Obviously, the caregivers could use the same device if they want, via Wifi, bluetooth or phone.
With this simple technology, we will reduce the incidence of SIDS, and will give peace of mind to parents.
Video
Like this entry?
-
About the Entrant
- Name:Diego Delia
- Type of entry:individual
- Patent status:patented