Chronic wounds that fail to heal quickly, such as diabetic foot ulcers or pressure ulcers, can be very tricky to manage for healthcare professionals and affect the life quality of patients. The treatment strategies are not only a healthcare burden but also present a challenge as early symptoms of developing wounds are difficult to ascertain. To monitor healing processes and assess the need for treatment, doctors and nurses normally need to remove the bandages from a wound, which harms the recovering tissue, often hurting the patient and requiring frequent hospital visits to avoid further developing infections. Furthermore, if wound inspection requires more than just visual examination, using other methods including tissue biopsies, surface swabs, or testing for pathogenic bacteria. These approaches are invasive and costly procedures that can take days and yet fail to produce useful treatment protocols. However, the development of ‘smart’ bandages, which are essentially wearable sensors that can monitor certain biomarkers during the wound healing process, have captured the attention of medical professionals.
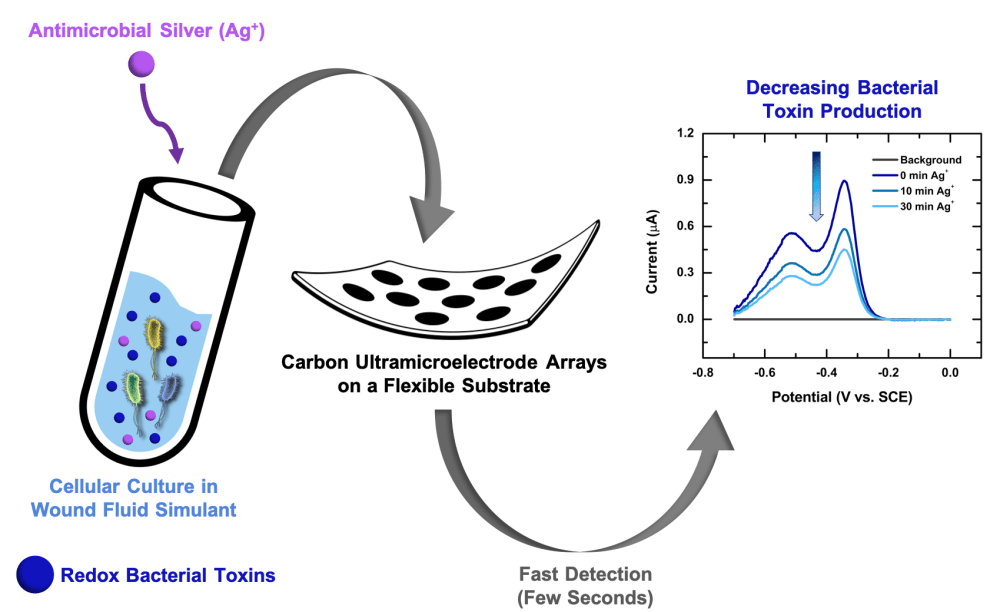
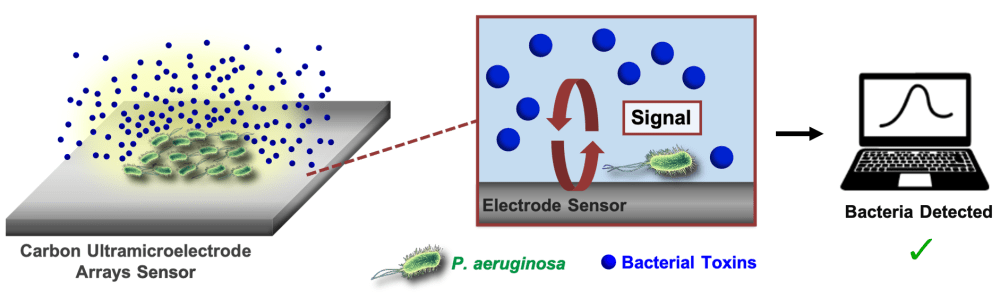
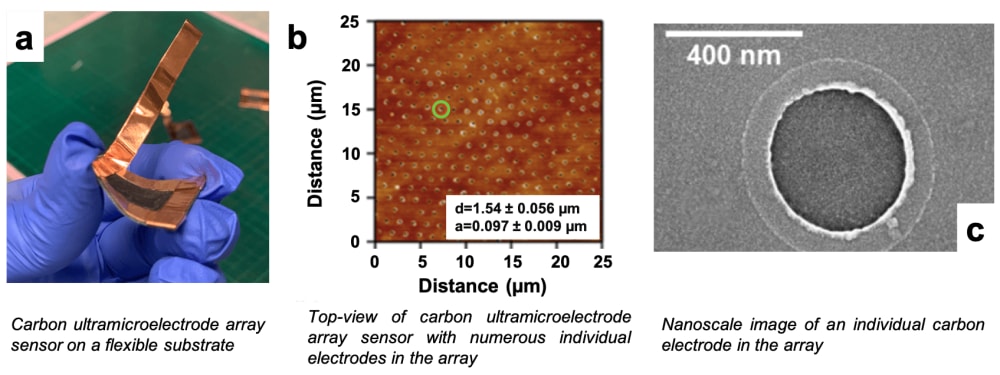
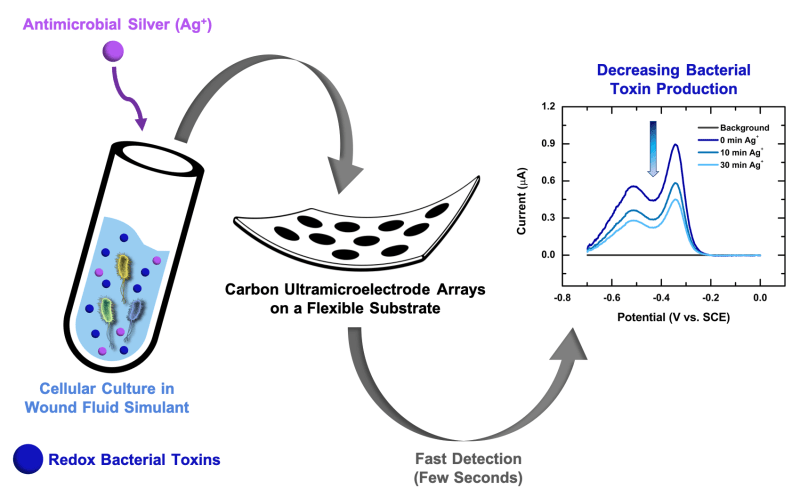
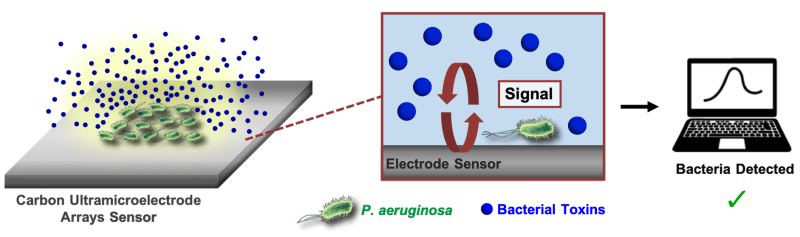
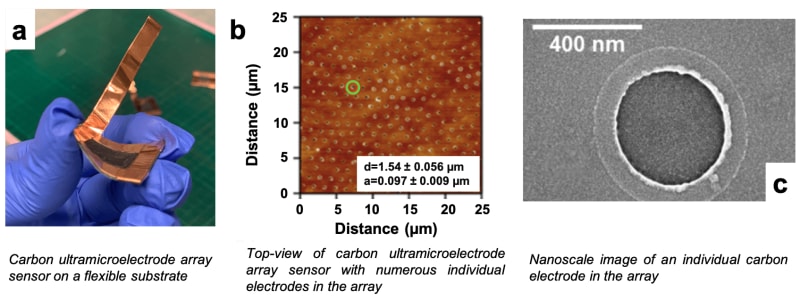
Electroanalytical methods are particularly promising for clinical applications due to their relative simplicity, sensitivity, durability, and other attractive characteristics. Our team created a prototype of a wearable electroanalytical sensor, based on carbon ultramicroelectrode arrays (CUAs) on flexible substrates, to monitor chronic wound activity. These low-cost devices (~ $12 per sensor) can quantitatively measure the state of wound healing, in real-time, and provide a ‘smart’ dressing to deliver wound treatment. The sensors are designed via a unique combination of carbon and metal oxide materials, resulting in nanometer sizes and array-based geometry that provide enhanced sensitivity for sensing in complex biological samples. Additionally, CUAs provide rapid detection (~seconds) and continual monitoring of wound healing state and infection by chemically tracking critical signaling molecules in chronic wounds.
A simulated wound environment was used to test the sensitivity of CUAs to three critical chronic wound biomarkers: (1) pyocyanin, a toxin molecule produced by Pseudomonas aeruginosa, a highly infectious bacterium typically colonizing chronic wounds, (2) nitric oxide secreted in response to bacterial infections by cells of the immune system, and (3) uric acid, a metabolite which strongly correlates with the severity of a wound. All these compounds are electroactive; they respond to electrical activity that can be detected by the electroanalytical sensor. The characterized CUAs limit of detection and linear dynamic ranges, namely the concentration ranges where a sensor produces meaningful quantitative results, are within the biologically relevant concentrations. The sensor was also tested with cell cultures to successfully detect pyocyanin from P. aeruginosa and nitric oxide from macrophages (immune cells that destroy bacteria and other ‘invaders’). Finally, the sensor detects the influence of silver (Ag+) ions, a well-known antimicrobial agent, that suppresses pyocyanin production by the bacteria.
Unlike other ‘smart’ bandage devices, our platform demonstrates sensitivity and selective detection of various analytes in complex wound fluid stimulants closely mimicking real biological environments. Thus, these engineered sensors have the potential to be a simple and effective solution in the treatment, monitoring, and healing of chronic wounds in clinical settings.
-
Awards
-
 2021 Medical Honorable Mention
2021 Medical Honorable Mention -
 2021 Top 100 Entries
2021 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Olja Simoska
- Type of entry:teamTeam members:[1] Keith J. Stevenson – Professor and Provost, Skolkovo Institute of Science and Technology (email: K.Stevenson@skoltech.ru)
[2] Olja Simoska – ACS Irving S. Sigal Postdoctoral Fellow, University of Utah (email: olja.simoska@utah.edu) - Software used for this entry:Autolab software
- Patent status:none