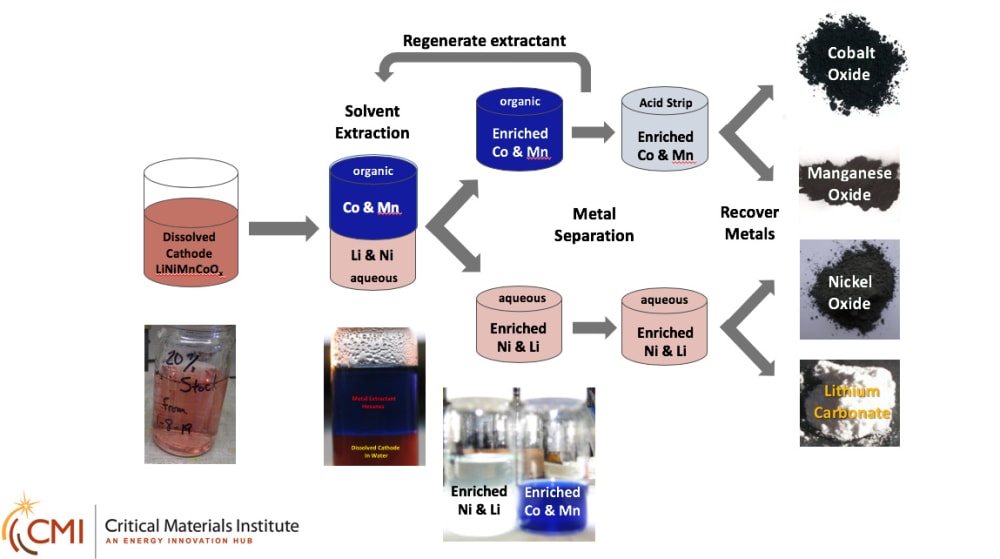
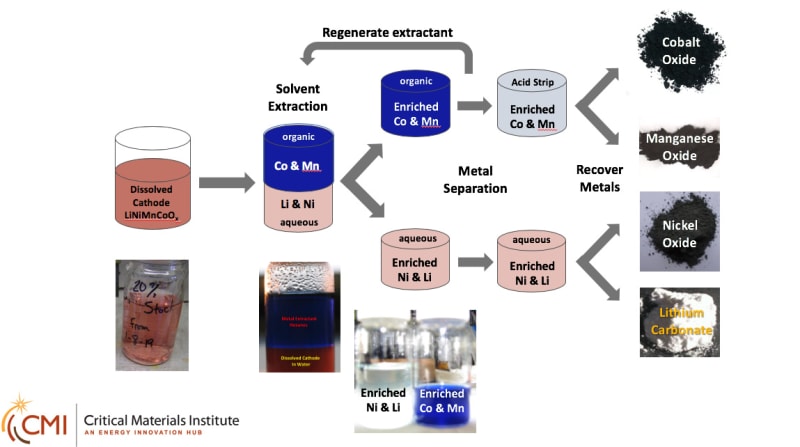
This process is an electrochemical process that effectively leaches cobalt and other valuable constituents from Li-ion battery electrodes. A reductant is used to solubilize the metal species in a dilute acid solution. Typical recycling process reactions usually require elevated temperatures and multiple hours for completion. INL’s process helps lower the cost of the recycling process. It uses inexpensive materials and can easily be scaled. It also uses less input chemicals. Both of these translate to lower cost and energy usage.
Researchers at INL have developed a process that effectively leaches cobalt and other valuable constituents from Li-ion battery electrodes at room temperature with short reaction times. This method has been extended to an electrochemical process which minimizes the amount of reducing agent needed and provides an inexpensive and scalable process for the cobalt recovery. The lower acid content is compatible with known metal extraction conditions needed for multi-metal battery chemistries (see attached figure).
-
Awards
-
 2021 Sustainable Technologies/Future Energy Honorable Mention
2021 Sustainable Technologies/Future Energy Honorable Mention -
 2021 Top 100 Entries
2021 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Andrew Rankin
- Type of entry:teamTeam members:Tedd Lister, Luis Diaz Aldana
- Patent status:pending