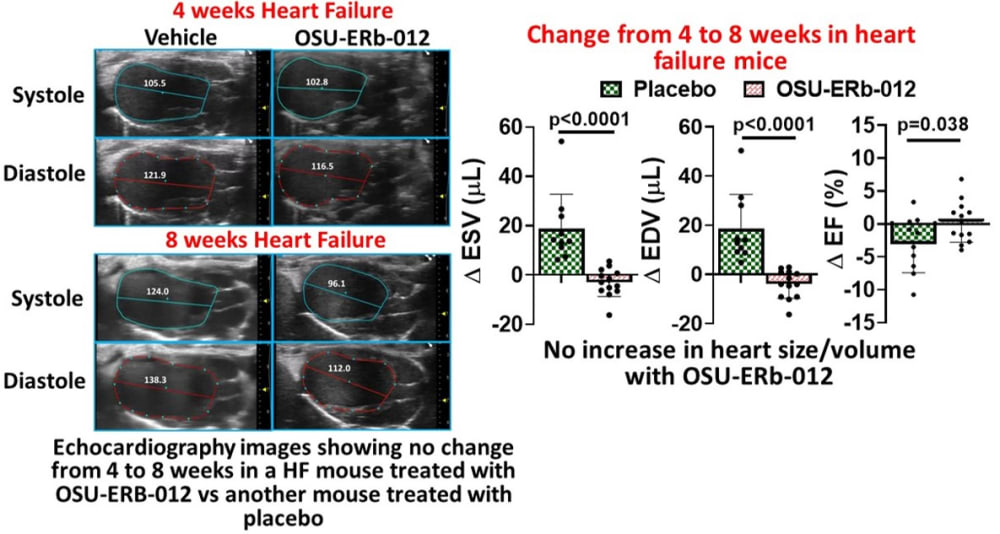
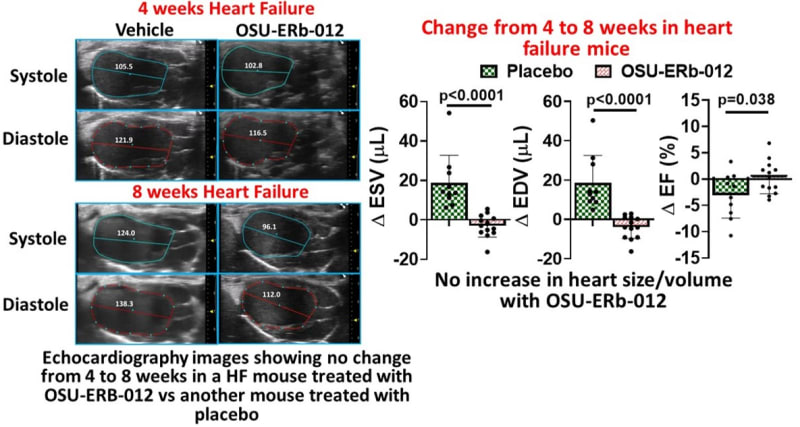
Current treatments for ischemic heart failure (HF) involve drugs that reduce cardiac load to facilitate heart function and there are no treatments available that can stop the disease from progressing. As a result, 50% of HF patients die within first 5 years. Our research of 10 years has shown that CD4+ T-cells, one of the immune cells that mediate wound-healing, become pathological during HF, and play a critical role in promoting cardiac remodeling and disease progression. More recently, we found that specifically during HF estrogen receptor (ER) α is activated in these pathological T-cells. ERα is a receptor that mediates the effects of estradiol, a hormone mostly present in females. However, it has been observed that ERα levels are increased in the hearts of male and female HF patients suggesting that ERα might be playing an important role in mediating this disease in a sex-independent manner. ERα effects are opposed by ERβ activation. Thus, to antagonize ERα, we identified a novel drug (OSU-ERb-012) that activates ERβ and found that this drug dose dependently inhibited the activation and proliferation of pathological T-cells isolated either from male or female mice (IC50 3.4 μM). To assess in-vivo efficacy of OSU-ERb-012, we induced heart failure in male and female mice and treated them with either placebo or with a daily oral dose of 60 mg/kg drug. During the treatment period, placebo treated HF mice showed progressive cardiac remodeling with significantly increased cardiac dysfunction similar to human patients (see illustration). However, treatment with our drug drastically diminished these changes by stopping progressive cardiac dysfunction and reducing cardiomyocyte hypertrophy and interstitial fibrosis. In addition, OSU-ERb-012 treatment specifically inhibited circulating and cardiac CD4+ T-cells in the HF mice, when compared with the vehicle treated HF mice, without altering other immune cells.
Our work shows that OSU-ERb-012 could be used as a selective immuno-modulatory treatment to inhibit CD4+ T-cells activated during chronic HF. This is the first drug that can stop disease progression in HF patients and could significantly increase their quality of life and increase patient survival.
-
Awards
-
 2022 Top 100 Entries
2022 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Shyam Bansal
- Type of entry:individual
- Profession:
- Shyam is inspired by:My passion to develop new drugs for heart failure and other cardiovascular diseases.
- Patent status:pending