ABSTRACT
We present a conceptual design, associated components, and method of assembling a paradigm shifting implantable artificial kidney system. This relatively simple device allows continuous, silent, and autonomous separation and removal of selected contaminants in a patient’s blood without using traditional filters and without application of external hydrostatic pressures. It has no moving mechanical parts, requires little power, and may be readily miniaturized and implanted in patients suffering from kidney failure for optimum, continuous, blood purification. This filtration/purification device relies on the patient’s own blood pressure. After passing through this device, cleansed blood is returned to the patient’s own circulatory system. Filtrate removed from this device is led into the patient’s own bladder for natural elimination from the body. The system does not require administering anticoagulants to prevent blood clotting.
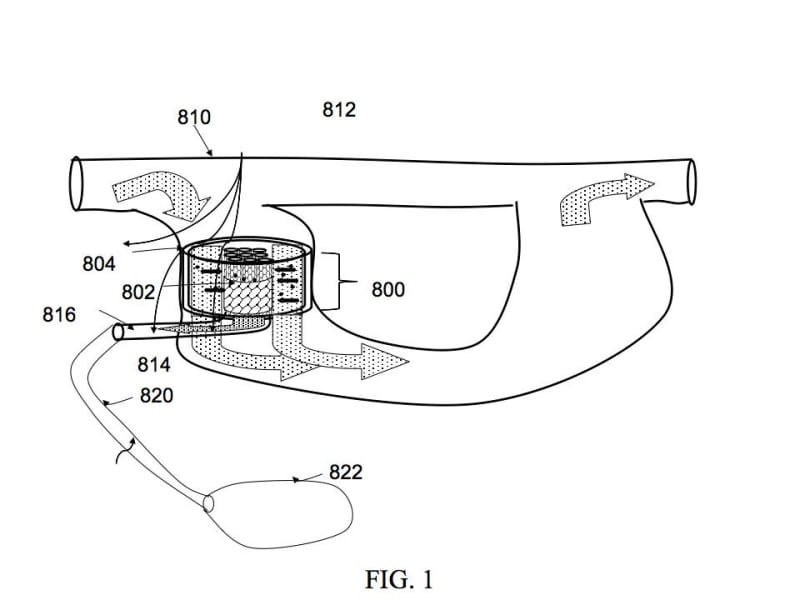
At the heart of this system is the use of tiny regulated electric fields to induce and control selective ion transport across semi-permeable nanotube walls. These nanotubes are enclosed within a larger, outer tube through which the patient’s own blood flows. Blood flows through the outer cylinder and travels alongside the length of these nanotubes. It is important to note that our system does not utilize traditional filtration properties of nanotubes. Blood is not purified by allowing it to pass perpendicularly across a membrane. Instead, in our novel design, blood flows in the outer tube alongside (i.e. parallel to) the enclosed nanotube length. In such flow patterns, very little purification of the flowing blood would be expected. However, when an electric field is applied across the outer tube, dissolved impurities in the flowing bloodstream (outside the nanotube) will be preferentially expelled from the flowing liquid stream and forced (directed) into the nanotube itself.
A tiny bias voltage (sub-millivolts) is applied across the outer tube (and the contained inner nanotube). A microprocessor carefully controls and regulates these applied voltages based on ion selective and/or protein specific sensors that monitor levels of various critical charged species in incoming and outgoing blood. In response, applied potentials and their on/off duty cycles across these tubes are varied. By regulating potentials applied across these tubes, high transport selectivity and preferential elimination of selected types of charged species in the bloodstream may be achieved. These hydrated charged species are driven inside these semi-permeable nanotubes where they form a separate waste stream. Toxins in the blood, such as salts and various unwanted low molecular weight molecules may be selectively eliminated.
Clotting of blood is prevented by means of specific anti-coagulating agents chemically anchored along those surfaces that contact passing blood.
Like this entry?
-
About the Entrant
- Name:Zvi Herschman
- Type of entry:teamTeam members:Zvi Herschman, MD, Miriam Herschman, BA, Yehuda Herschman, BA
- Patent status:pending