Diseases of the central nervous system (CNS) in humans are still very complex and remain a vastly unsolved challenge for medical and pharmacological research. Even in today’s world, a lot of pathological research for CNS diseases are conducted on laboratory animals. But limited translational significance from animal to human model, and high costs have driven a need for the development of in-vitro models. One area of particular interest is the in-vitro replication of cerebrovascular systems in the human brain. The blood-brain-barrier (BBB) is a filter system made up of tightly packed endothelial cells that perform a complex gatekeeping job.
Understanding the mystery behind the pathways of the BBB can help ease drug delivery, and timing of drug delivery for treatment of CNS diseases. We already know that certain molecules which have lipid affinity, others such as alcohol can pass through easily. Mimicking the BBB can also help predict failure scenarios of the BBB, which are one of the early predictors for Alzhemiers, epilepsy, multiple sclerosis, etc.
We aim to develop a cost-effective, scalable microfluidic device simulating the blood-brain barrier (BBB) at the cellular and molecular levels. Microfluidics is a branch of engineering that studies fluid flow at very small length scales and narrow channels and it is well suited for study of human microcapillaries. Microfluidic models have been developed previously to recreate the BBB in some fashion or another but they all lack overall chemical complexity and the necessary accuracy to be compared to the human BBB.
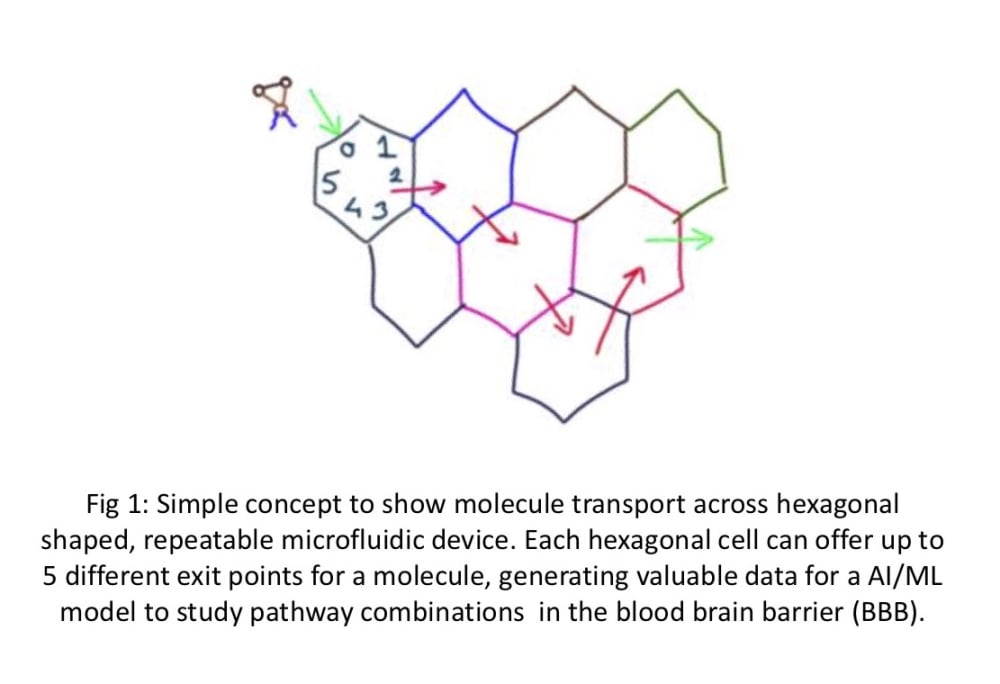
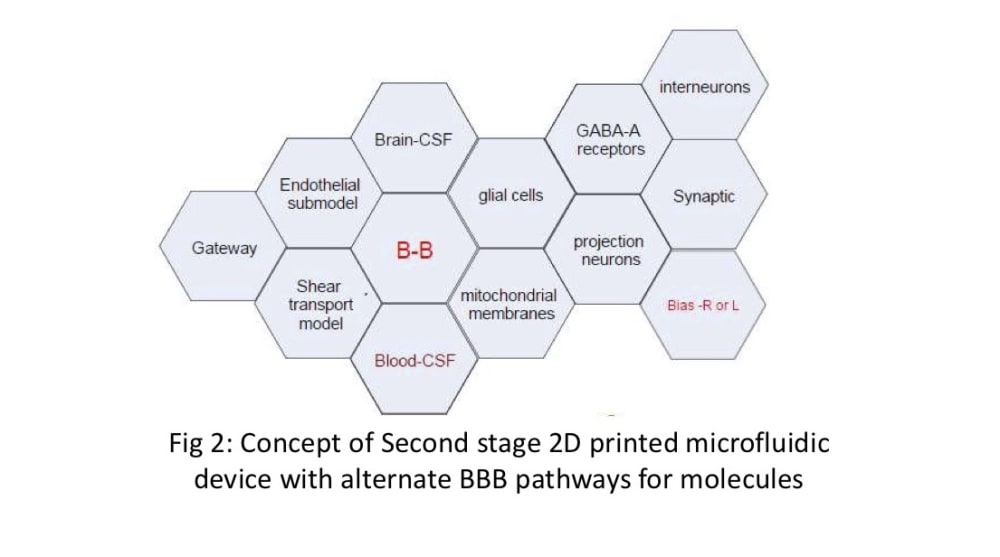
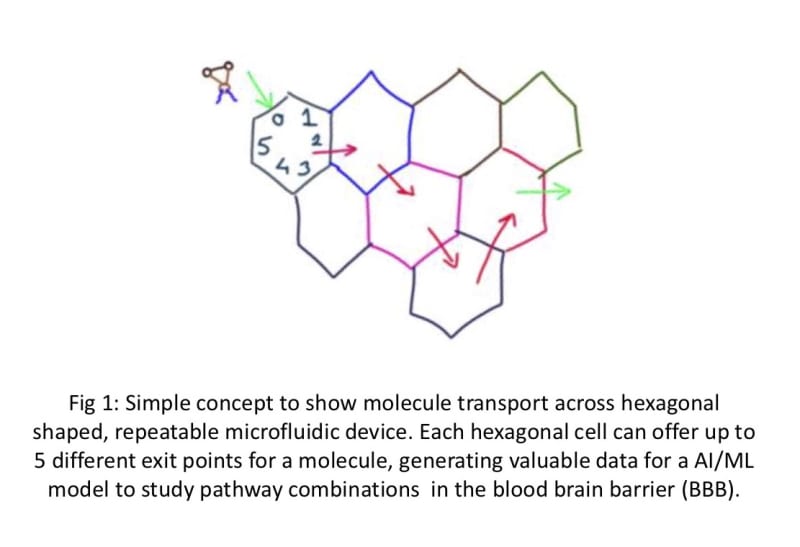
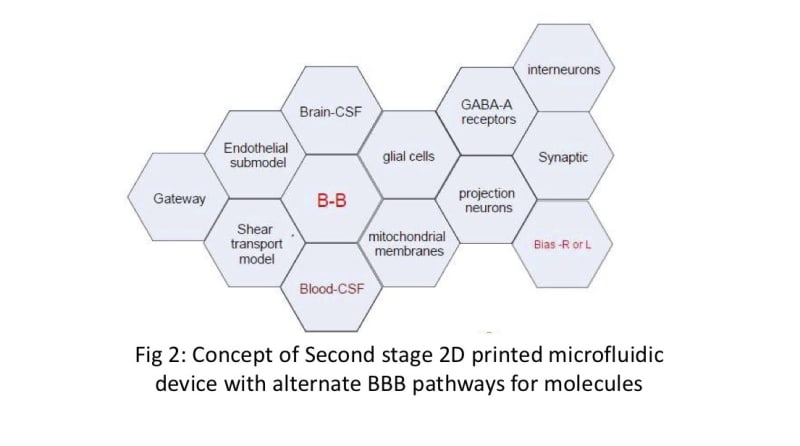
We propose a novel 2D beehive model to create and test the physico-chemical gating capabilities of the BBB. The use of individual hexagon-shaped cells joined together in a customizable 2D-beehive like network enables ease of scaling up and manufacturing using available printing (3D printing), coating and assembling techniques. Each “cell” can offer 4-5 up to different passage options and in combination with other cells in the “hive” network, offer a manageable way to iteratively study a large degree of combinations of cell wall qualities. Data generated from one test run can be used for Machine Learning (AI/ML) and help manufacture the next iteration of microfluidic cells with the proper gate combinations. Individual cells can be turned off or turned as needed in this circuit.
We are building on current work that uses DIV-BBB and NDIV-BBB (rat) models by exploring the shear flow with glial cells mimics. Our methodology includes deactivating glial cells in microfluidic cultures, assessing organ behavior, and using impostor glial cells to study signal transport. Synaptic function will be manipulated through local sucrose loading. Electrical resistance measurements will evaluate BBB layer elasticity. Our goal is to enhance customization on the "blood-brain barrier on a chip" and create carrier-receptor systems using extracts of medicinal plants.
In conclusion, this device will contribute to understanding neural degenerative diseases and potentially enable active drug delivery for psychiatric conditions.
-
Awards
-
 2023 Top 100 Entries
2023 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Narasimhan Soundarrajan
- Type of entry:individual
- Profession:
- Number of times previously entering contest:1
- Narasimhan's favorite design and analysis tools:Google Sketchup
COMSOL Multiphysics (when license available)
DWSIM (open source) - Narasimhan's hobbies and activities:Nature photography, bird watching, cooking
- Narasimhan is inspired by:A lot of my design thoughts are usually inspired by extreme environments in nature and how many creatures adapt to those environments. Biomimicry is the most sincere form of flattery we could ever do to give respect to our environment!
- Software used for this entry:None
- Patent status:none