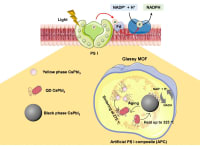
The industrial use of enzymes for chemical production currently represents a 100 billion USD industry. A major application of enzyme catalysis is enantioselective synthesis in the pharmaceutical sector, with two-thirds of commercial chiral products involving enzyme catalysis. Up to 25% of known classes of enzymes require costly stoichiometric cofactors such as nicotinamide adenine dinucleotide (NAD+) and its reduced form (NADH). However, cofactor NADH regeneration in the industry is currently performed with relatively low productivity and a costly separation process. Therefore, it would be more viable to regenerate it in an economically feasible and environmentally sustainable manner. In this scenario, solar-driven photocatalytic production of NADH is extremely promising, offering a way to mimic the natural photosynthesis process.
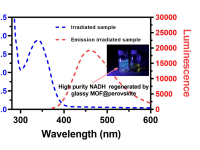
Our team brings the selectivity and sufficiency problems of NADH regeneration into focus and aims to develop a novel photocatalyst for commercially viable NADH production. In the past decade, research into the use of perovskites as photocatalysts has become increasingly popular due to their desirable band-gap structure and remarkable availability. However, perovskite's instability is the primary obstacle to achieving the desired performance. Within this context, our team recently designed a novel heterojunction photocatalyst consisting of Rh-doped glassy MOF (Rh-agZIF-62) and nanoconfined perovskite (CsPbI3), in which the active phase of perovskite can be stabilized at room temperature. Therefore, electrons can then be generated by photoexcitation of active perovskite crystals and then transferred through the glassy MOF’s electron mediator (i.e. Rh site) for effective NADH regeneration. Up to 80% NADH yield could be achieved by our heterogeneous photocatalyst upon light irradiation.
-
Awards
-
 2023 Top 100 Entries
2023 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Wengang Huang
- Type of entry:teamTeam members:
- Jingwei Hou
- Patent status:none