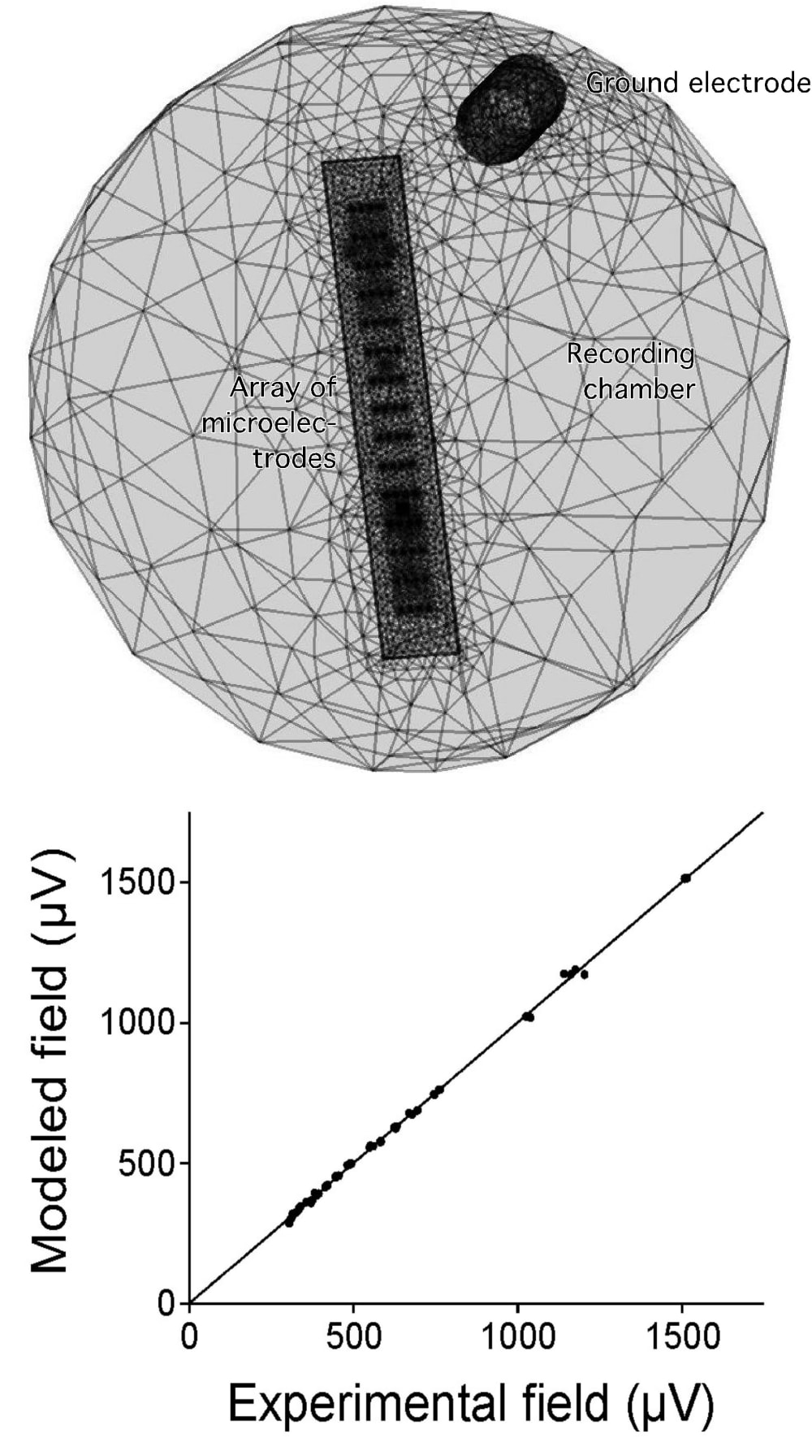
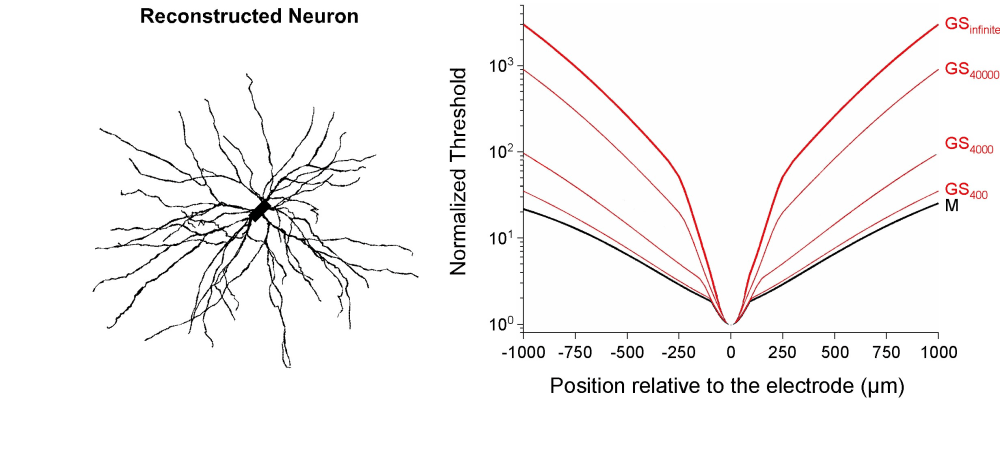
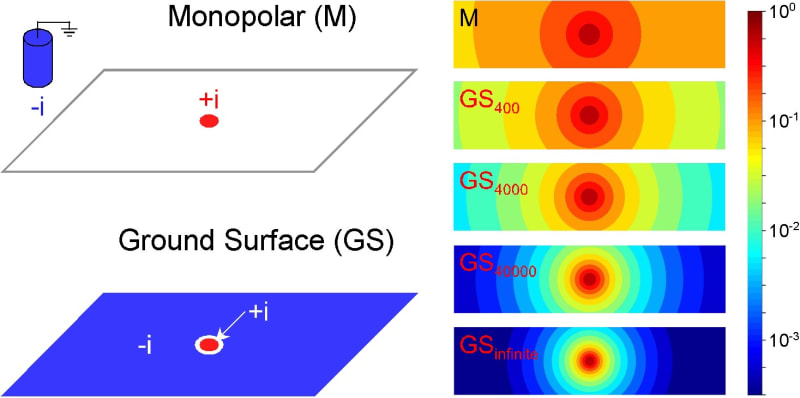
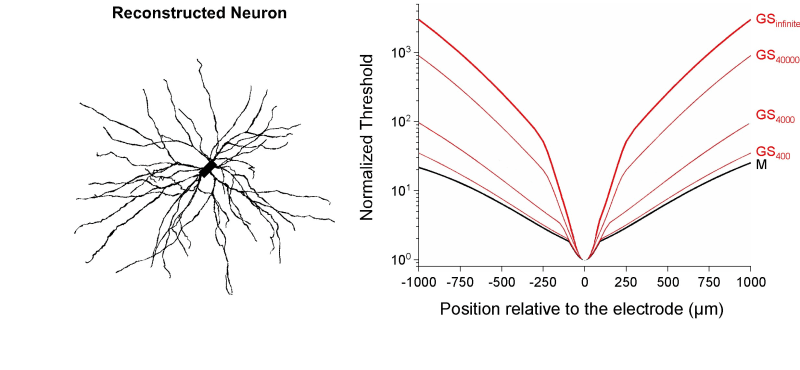
Currently, more than 100 million people suffer from neurodegenerative diseases worldwide (e.g., 30 millions from macular degeneration of the retina, 25 millions from Alzheimer, 6.5 millions from Parkinson), and the global population ageing will be accompanied by an increase of patients. Among treatments for the rehabilitation of neural functions, neuroelectronic strategies making use of electrical neural stimulation are increasingly considered as future healing routes. These include neural implants and prosthesis, where arrays of microelectrodes (MEAs) combined to integrated microelectronics are implanted in the Central Nervous System (CNS). Electrical stimulation of the CNS has been used empirically for decades, with both fundamental and clinical goals. One salient success is the treatment of tremors in Parkinson patients using macroscopic electrodes implanted in the brain. Yet, the underlying mechanism and the spatial extent of stimulated tissue are not well understood and controlled. The development of MEA technology now offers new routes toward more precise and locally controlled stimulation paradigms using microscopic electrodes. This is for instance of great interest for the development of retinal prosthesis to compensate macular degeneration: arrays of microelectrodes are implanted on the surface of the retina to restore vision using precise spatio-temporal electrical stimuli mimicking images of the outside world. In order to achieve well-defined stimuli, one challenge is to build electrode arrays allowing focal electrical stimulation, yielding activation of cells only in the vicinity of each microelectrodes. In this perspective, we developed a new electrode configuration allowing focal electrical stimulation. First, we validated a Finite Element Model to compute the potential field generated in a neural tissue by an electrical stimulation delivered by MEAs. Unlike previous models using classical Dirichlet boundary conditions, this model uses Robin boundary conditions that take into account the surface conductance of electrode/medium interfaces. A direct comparison between calculated and experimentally measured potential values confirmed the accuracy of the model and the importance of modeling the electrode impedance (Figure 1). Then, using this model, we evaluated the focality of the potential field for different electrode configurations. We also combined a compartment cable model of realistic neurons to compute the electrical response of a neuron to the applied field and determine how the minimal current required to activate the cell (threshold current) varies with distance (current-distance curves) for the different electrode configurations. We propose a new electrode configuration to improve the focality of both the potential field (Figure 2) and the current-distance curves (Figure 3), which consists in using a ground surface (GS) surrounding all the electrodes of the array for the return of the stimulation current. We show that the higher the interface conductance of this surface, the more focal the stimulation. In particular, using Neuman In conclusion, this study proposes new boundary conditions for the design of precise computational models of neural stimulation, and a new electrode configuration that can be easily incorporated into future MEA devices to achieve better spatial control of CNS microstimulation. Such approach should help building more efficient neural prosthesis to restore functional activity in the injured CNS.
Like this entry?
-
About the Entrant
- Name:Blaise Yvert
- Type of entry:teamTeam members:Sébastien Joucla Blaise Yvert
- Hardware used for this entry:Pentium IV 2.4 GHzSoftware used for this entry:Comsol Femlab 3.1a
- Patent status:patented