Alair aluminum-air battery for sustainable electricity production
This device is intended as a standalone electricity generator for off-grid living, in either developed or developing countries. It uses the chemical reaction:
4Al + 3O2 + 6H2O ? 4Al(OH)3
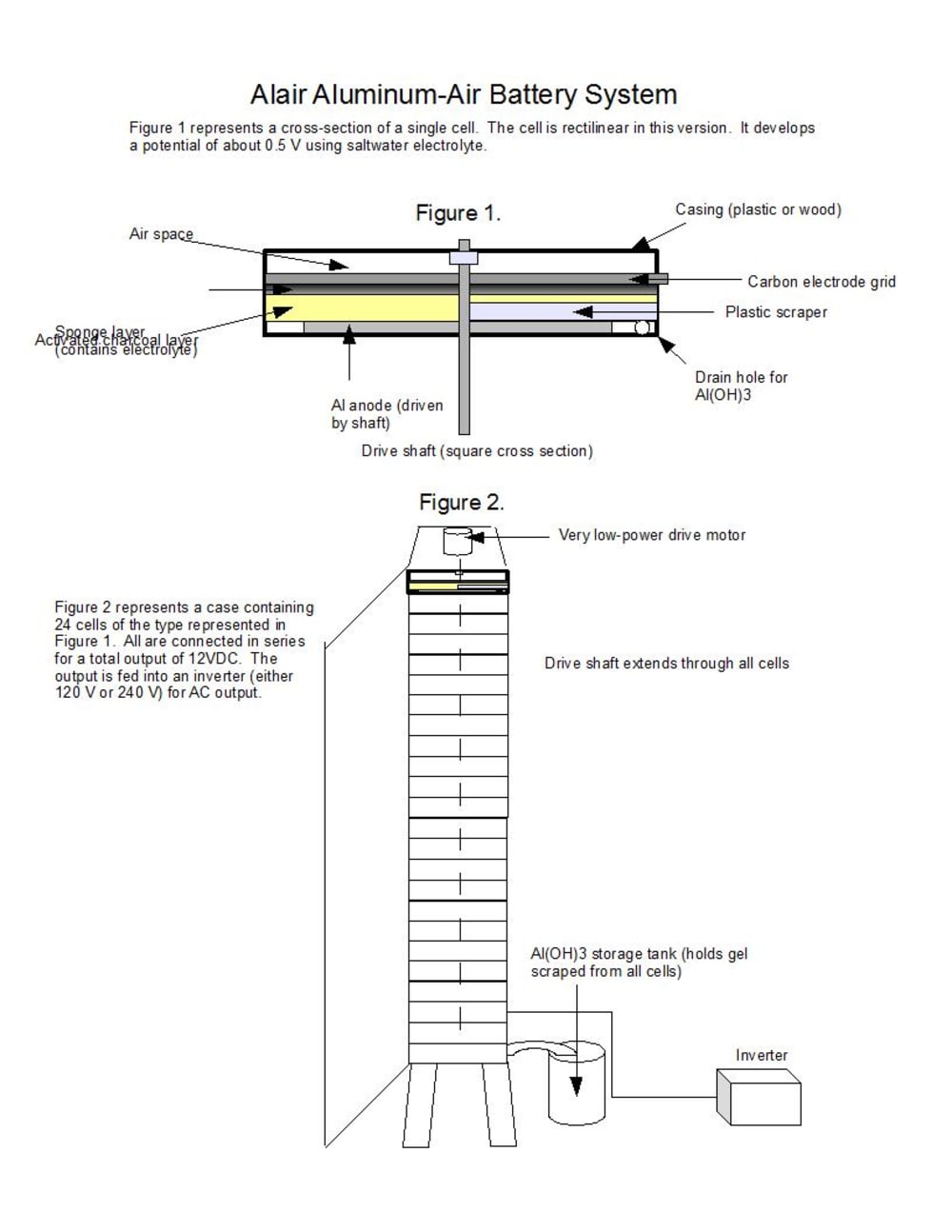
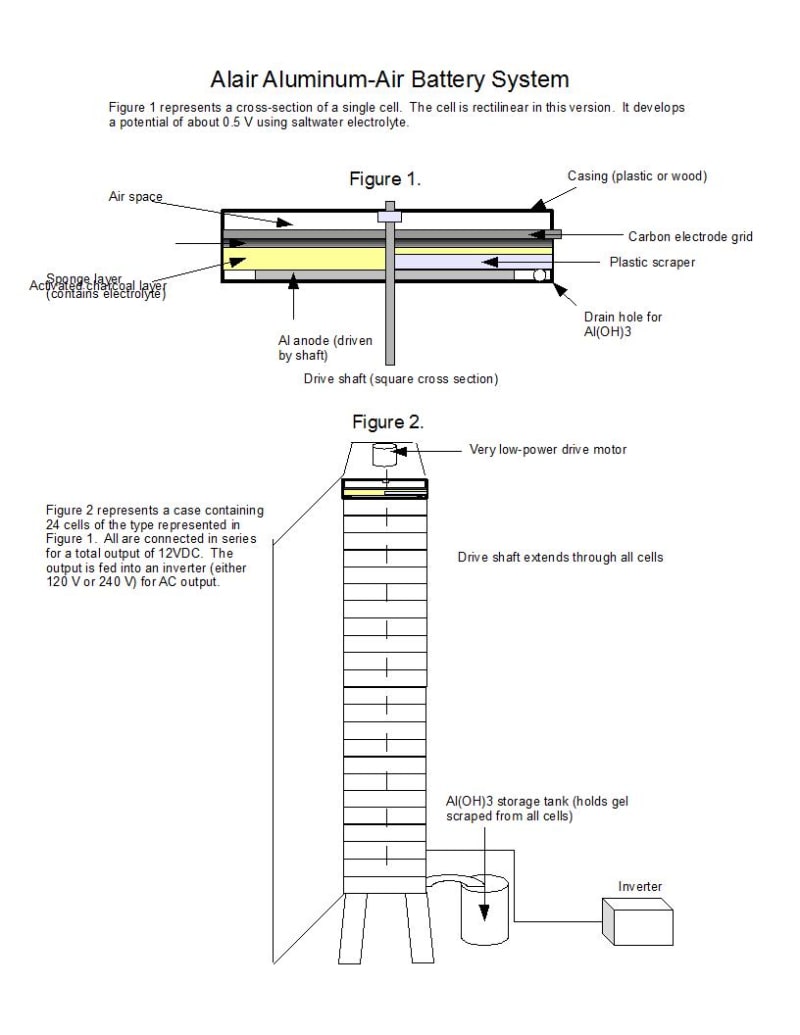
to produce 12 VDC electricity which is then inverted to produce either 120 VAC or 240 VAC output. The device is simple to manufacture and uses easily-obtainable materials. Salt water is used as an electrolyte, aluminum plates for anodes, and activated charcoal as the air electrode. A carbon waffle grid embedded in the charcoal serves to collect electrons. Sponge is used to contain the electrolyte. Since the saltwater reaction produces about 0.5 V, 24 cells are required.
One of the major problems with Al-air batteries is the buildup of aluminum hydroxide gel on the plates. The present design mounts all anode plates on a single shaft of square cross-section, via a slot cut in each plate. The shaft is driven by a low-power electric motor. Each plate may be installed and removed by hand simply by sliding it into or out of the cell. Each cell has a fixed scraper to remove aluminum hydroxide buildup from the plates. The aluminum hydroxide is channeled into a collection tube, then to a storage tank for recovery.
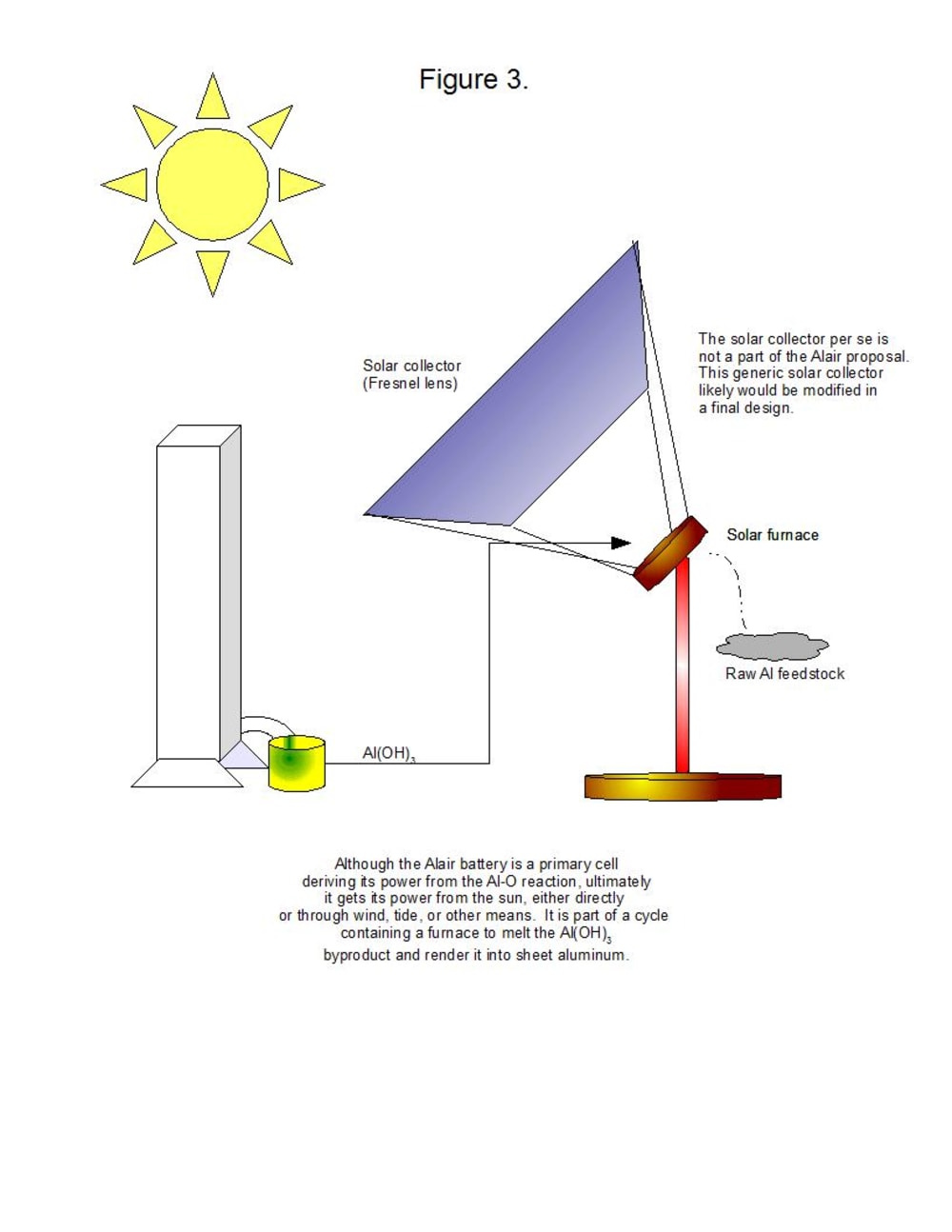
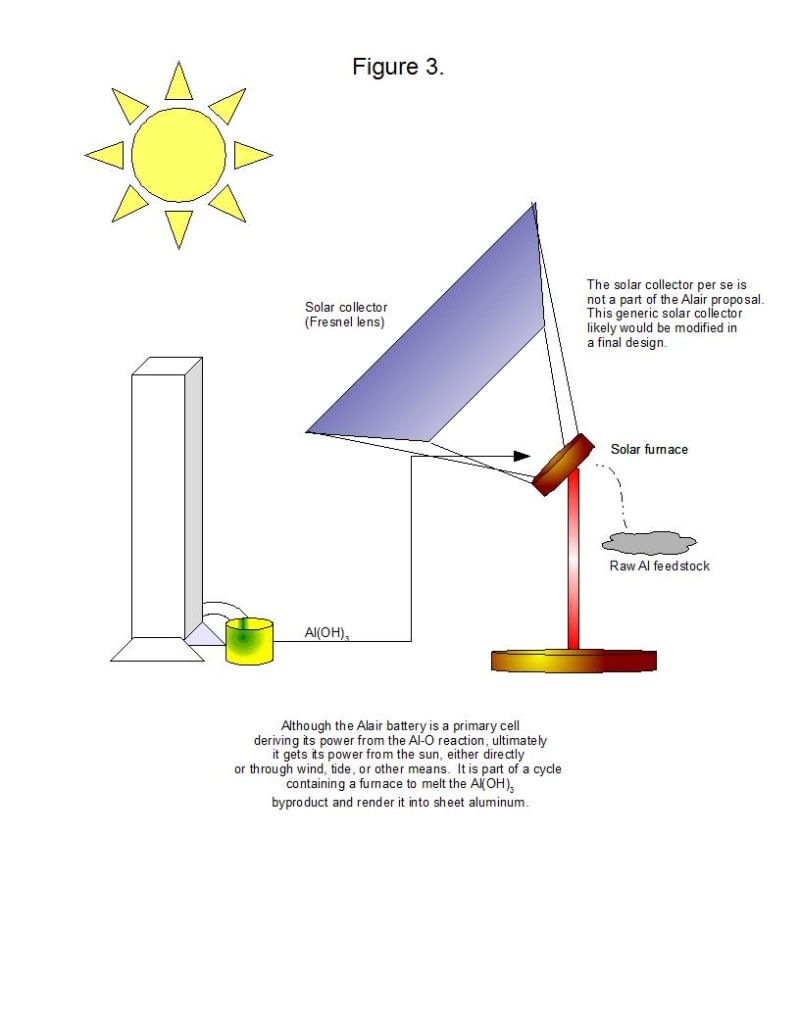
It should be noted that the aluminum anodes are consumed by the reaction, so that a constant supply of aluminum is required. The main product of the reaction, aluminum hydroxide, can be heated to 572 degrees F. (300 degrees C.) in a solar furnace to recover pure aluminum. Thus, the Alair battery can be part of a closed system that ultimately depends on the sun. Unlike pure solar power, however, the Alair battery can be used as a source of power at night and on cloudy days.
Aluminum hydroxide is a valuable product in itself, being the primary component of many antacids that sell for $6-9 USD per 16-oz. (454 g) bottle. It is possible that selling aluminum hydroxide wholesale or retail may be used as a way of offsetting the capital cost of the Alair battery.
The Alair battery is easily scalable from small to very large sizes. It uses aluminum for anodes, plastic or wood for the case and for cell dividers and scrapers, sponge, activated charcoal, carbon, and salt water. Dimensions are not critically important, and are determined by practical matters such as size and location of the space in which the battery is to be installed. Alair is possibly the cheapest battery by any measure to manufacture. It has the potential to greatly improve quality of life in areas where electricity is unreliable or absent, not to mention the side benefit of reducing dependence on fossil fuels.
Like this entry?
-
About the Entrant
- Name:Michael Flora
- Type of entry:individual
- Hardware used for this entry:Toshiba SatelliteSoftware used for this entry:Blender, OpenOffice Draw
- Patent status:pending