Including a phase change in a closed cycle heat engine is innovative because liquids expand so many times their volume when boiled. The working gas in a Stirling engine expands at a linear rate when heated creating the pressure changes needed. The pressure changes in a confined volume of gas will be much larger if a small quantity of liquid is along for the ride. The ideal format will also return the warm condensate to the starting point quickly.
Presented is a patent pending closed cycle heat engine that includes a phase change to leverage the transfer of thermal energy. The pressure change is multiplied by the phase change of the liquid during the thermal transfer. The reactions in each chamber move the rotor forward. Solar thermal, geothermal or waste heat recovery, this design is reliable, versatile, scalable and economical.
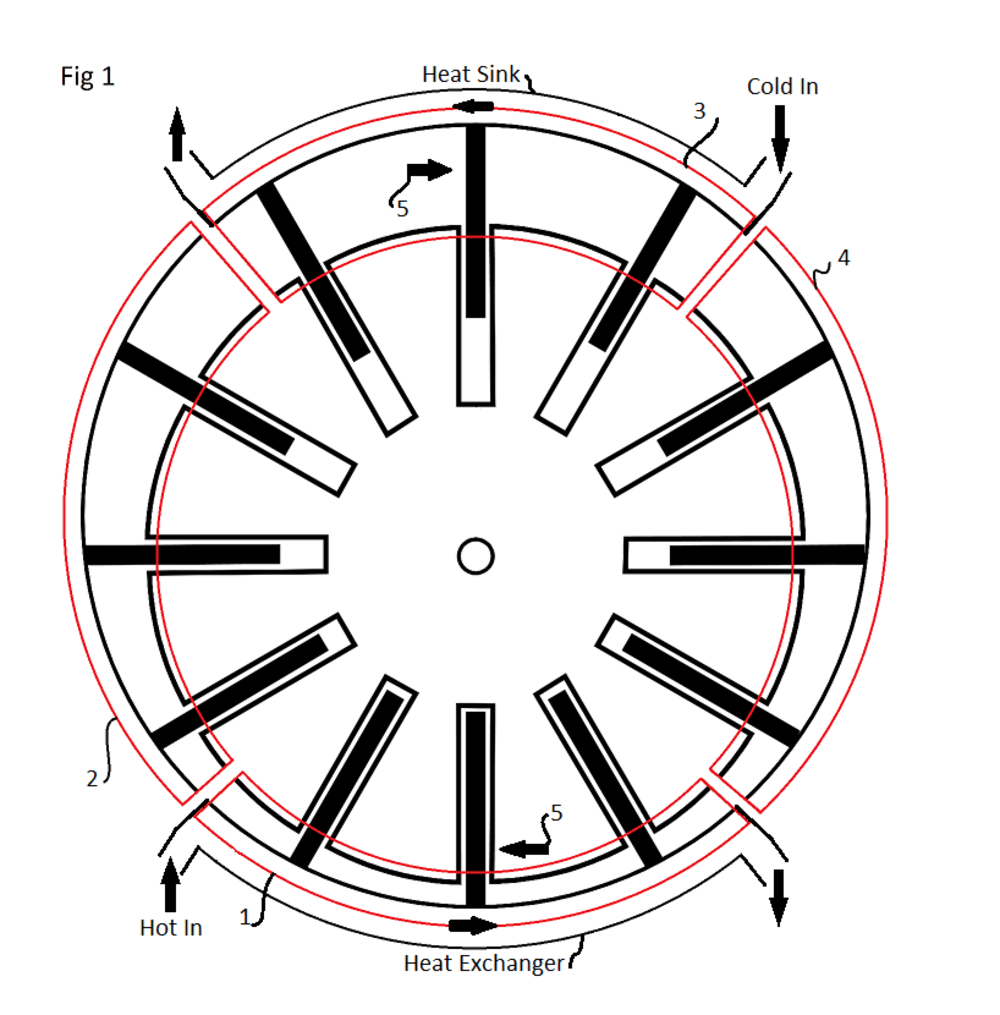
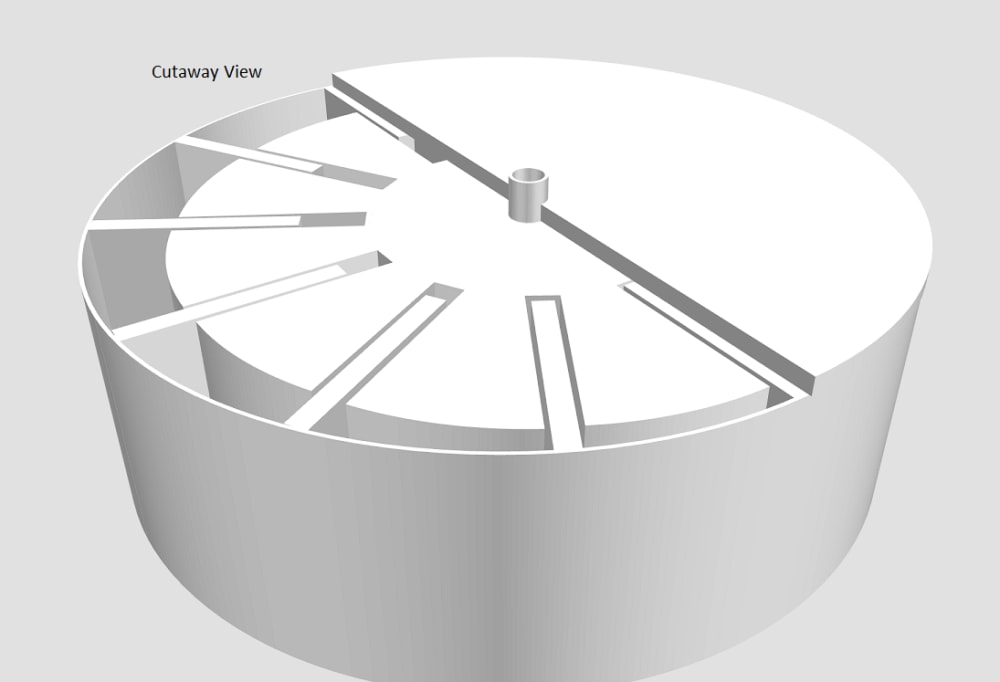
The sliding vane thermal exchange engine has a closed cylinder with a rotor having an offset axis and multiple sliding vanes attached (Fig 1). The chambers formed by the sliding vanes increase and decrease in volume on each rotation. The activity in the circular cycle of each chamber can be visualized when separated into quadrants (red outline), including two areas where the chamber volume changes very little, one large (3) and one small (1) and two where the chamber volume is either increasing (2) or decreasing (4) when the rotor is turned (clockwise 5).
Before starting the cylinder is closed, allowing the thermal energy to be transferred though the cylinder walls. Confined in the cylinder is a specified pressure of gas and a less than 3% by volume, quantity of phase change liquid. A gas such as helium is specified for its heat transfer coefficient and the liquid is specified for its boiling point at the pressure and temperature range anticipated.
The cycle starts by transferring thermal energy into a chamber adjacent to the quadrant having the small average chamber volume (1). The transferred thermal energy both expands the gas contained and causes the liquid contained to boil (change phase), increasing the pressure. The increased pressure pushes the chamber through the next quadrant, where the volume is increasing, (2) turning the rotor. As each chamber moves though the quadrant having the large average volume (3), the thermal energy is transferred out, causing the gas to contract and the boiled vapor to condensate, reducing the pressure contained. The reduced pressure will now cause the described chamber to move through the quadrant having a declining volume, (4) then back to the start, carrying with it the quantity of condensate ready to be expanded to vapor again.
Like this entry?
-
About the Entrant
- Name:Terry Vaninsberghe
- Type of entry:individual
- Software used for this entry:Paint
- Patent status:pending