The Problem: Prior to the development of cardiac phantoms, the development of myocardial imaging protocols required a natural heart. Heart phantoms model the fluid mechanics and functionality of the heart, allowing researchers and physicians to calibrate imaging equipment for modeling and protocol development. Traditional phantoms have been static, cost thousands of dollars, and are only compatible with a single mode of imaging (unimodal). With comprehensive system analyses for accurate, personalized healthcare necessitating multiple phantoms, there is a high probability of inconsistency in results and prohibitive costs in phantom development and acquisition. As multimodal imaging protocols continue to advance, the need for a multimodal calibration phantom will dominate the imaging phantom market.
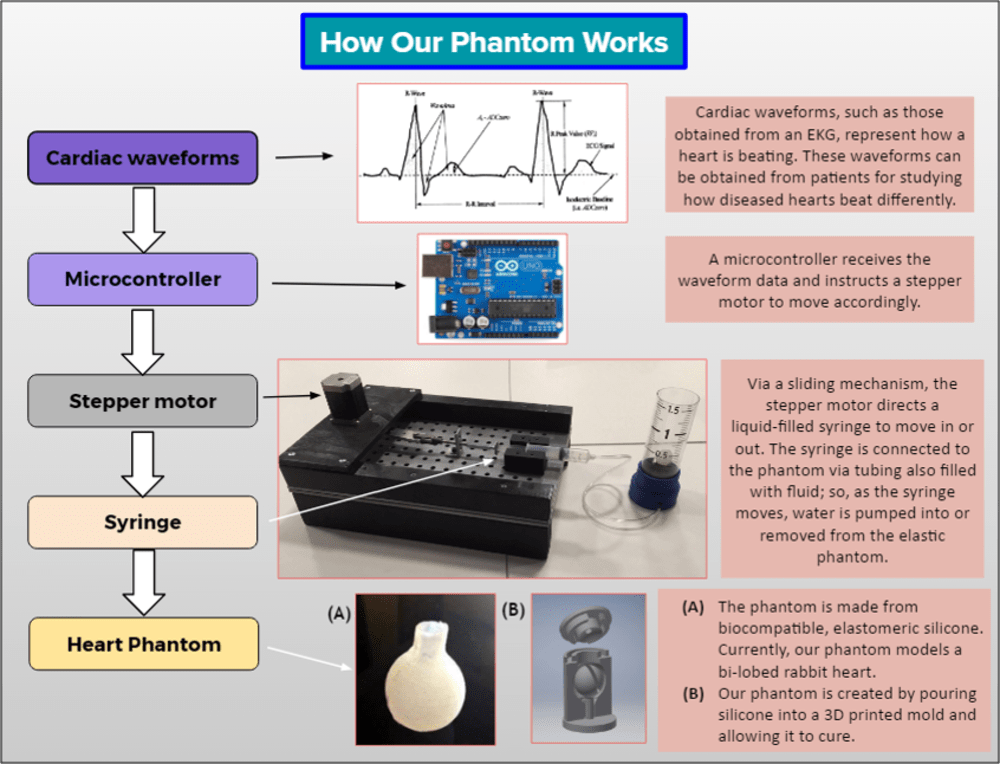
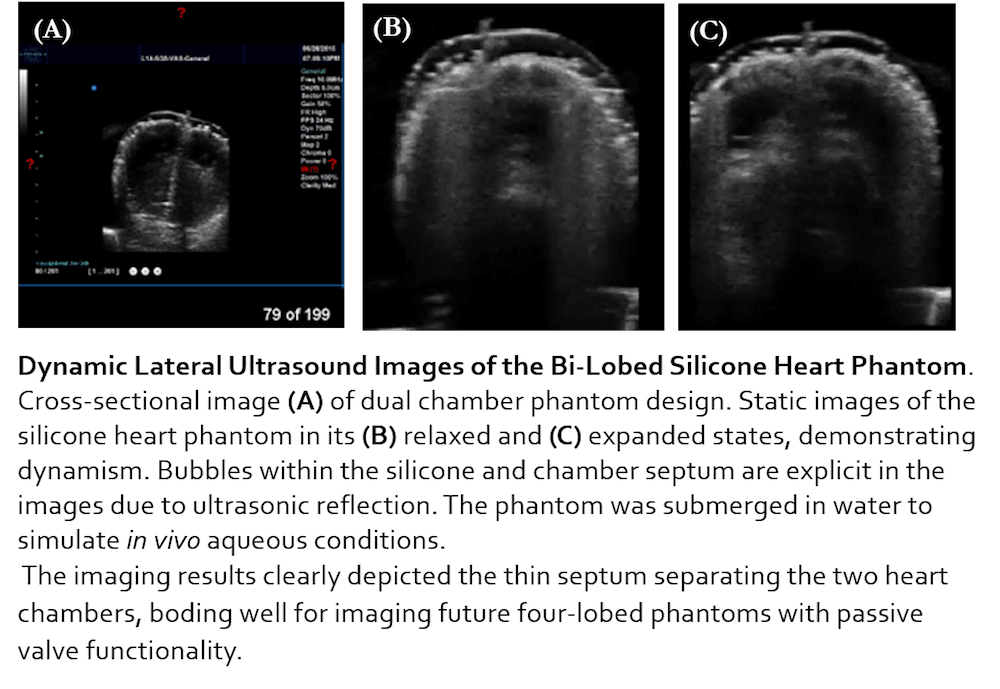
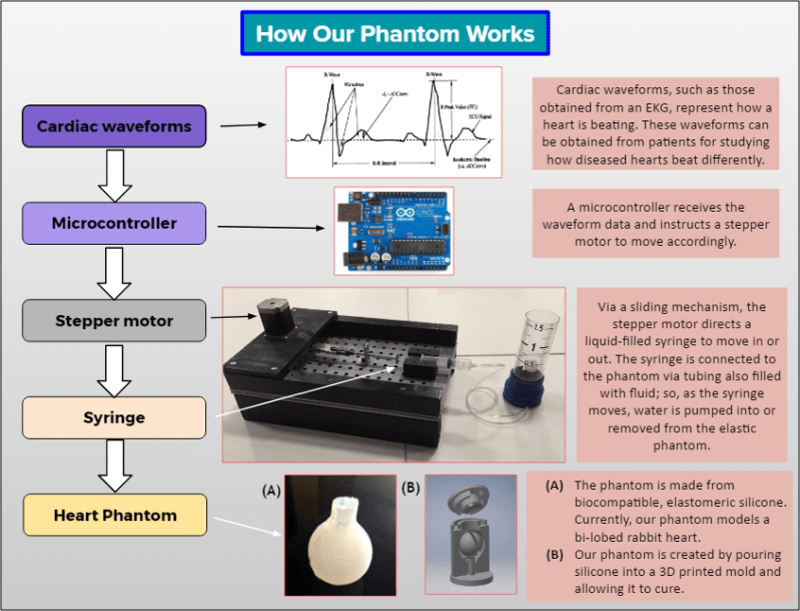
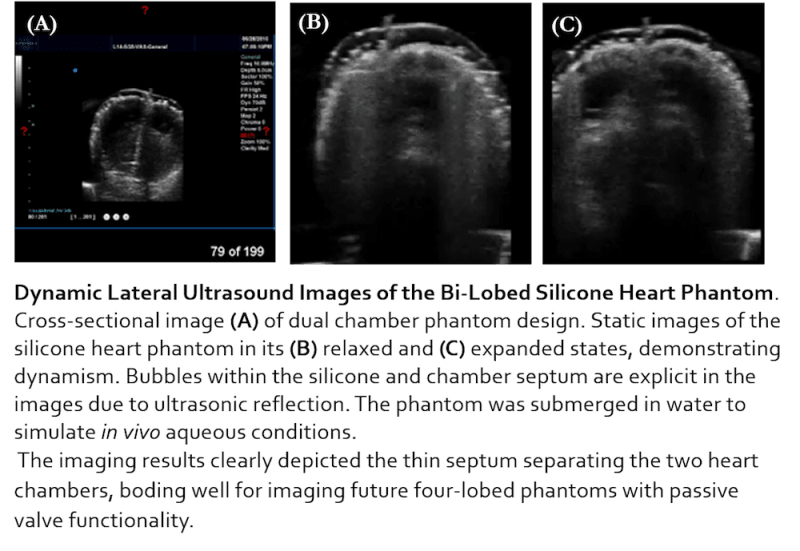
The Solution: PhantomCor is a dynamic, multimodal, and inexpensive heart phantom compatible with computed tomography (CT), magnetic resonance imaging (MRI), and ultrasound imaging. Fabricated from silicone chosen for its similarity to human tissue, the phantom is developed within molds 3D printed per the specifications and disease pathology of the heart being modeled; thus, phantoms can be rapidly prototyped for modeling any desired heart condition or disease state. The phantom is pumped using a syringe-pump system oscillating according to cardiac waveform data, and is currently prototyped to the dimensions of a bi-chambered rabbit heart. Several phantoms have been successfully fabricated, both single-lobed and bi-lobed in design, by generating 3D printed molds of rabbit heart models, and progress is being made to develop a human heart-sized model. Data obtained from CT, MRI, and ultrasound imagery using cardiac imaging protocol affirm the multimodality of our phantom and demonstrate its capability for modeling human tissue.
Market and Business Strategy: PhantomCor would sell at just $500 for the physical device and accompanying software, thus providing a much more affordable alternative to current market phantoms (costing several thousands of dollars). The initial market for PhantomCor is the medical device R&D market, a $7.3 billion total market as of 2011. Secondary markets include medical educators using the phantom to teach cardiac structure and fluid mechanics, hospitals using the phantom for personalized cardiac healthcare, and imaging institutions using the phantom for equipment calibration. In the US alone, there are 141 medical schools, 400 teaching hospitals, and over 25,000 MRI and CT machines, and the medical imaging market is projected to grow at a CAGR of 5% by 2020.
The apparatus falls under the electro-medical equipment classification of non-invasive medical devices and requires 510(k) Class II FDA approval, which will only take 6-8 months. PhantomCor units would be bought from our team by hospitals, medical imaging centers, medical education institutions, and research groups. Following the initial purchase, customers could request the fabrication of more phantoms for use with their device to simulate other heart pathologies (i.e. a medical researcher could request a phantom based off a patient’s CT scan); the PhantomCor team would fabricate additional hearts for $50 apiece.
Thus, PhantomCor offers an inexpensive solution to machine calibration and paves the road to more accurate and personalized healthcare, improved medical education, and novel medical protocol development.
Video
Like this entry?
-
About the Entrant
- Name:Hiba Shahid
- Type of entry:teamTeam members:Hiba Shahid
Joshua Au
Viraat Goel
Pierce Hadley
Alex Hasnain
Boeun Hwang
Monika Kizerwetter
Joshua Lew
Bara Saadah
Craig Soares
Linsun Sunny
Teresa Yang
Hugh Yeh - Patent status:none