Disease State: Heart failure and irregular heart rhythms (arrhythmias) result in about 1M people in the US implanted with pacing systems comprised of a pacemaker device and from 1 to 3 leads attached to the inside of the heart chambers or coronary vessels. The inherent blood-contacting nature of these endovascular leads and limitations in their placement results in over 10% complications – about $650M in healthcare costs - related to valve damage, heart wall perforations, heart failure from right ventricle pacing, ventricular dyssynchrony, endocarditis, inappropriate shocks, and lead failures. Traditionally-placed coronary vessel leads are limited by the locations of coronary sinus branches, their caliber, and the associated implantation challenges. Delayed left atrium activation from right atrium pacing reduces mechanical and electrical function of atria, possibly even leading to atrial fibrillation.
Current Solution: The current pacing solutions utilize primarily endovascular leads. New leadless pacemakers address infection and cyclical failure risks, but their indications for use are very limited. Furthermore, they are still endovascular, and their limited battery life will require frequent replacement with a risky interventional procedure. They also cannot pace the left side of heart. The epicardial leads that do exist fixate into the myocardium generally with an open procedure, and do not span multiple chambers.
Clinical Need Statement: There remains a clinical need for a less invasive heart pacing solution to reduce complications and one that provides improved atrial and ventricular synchronization.
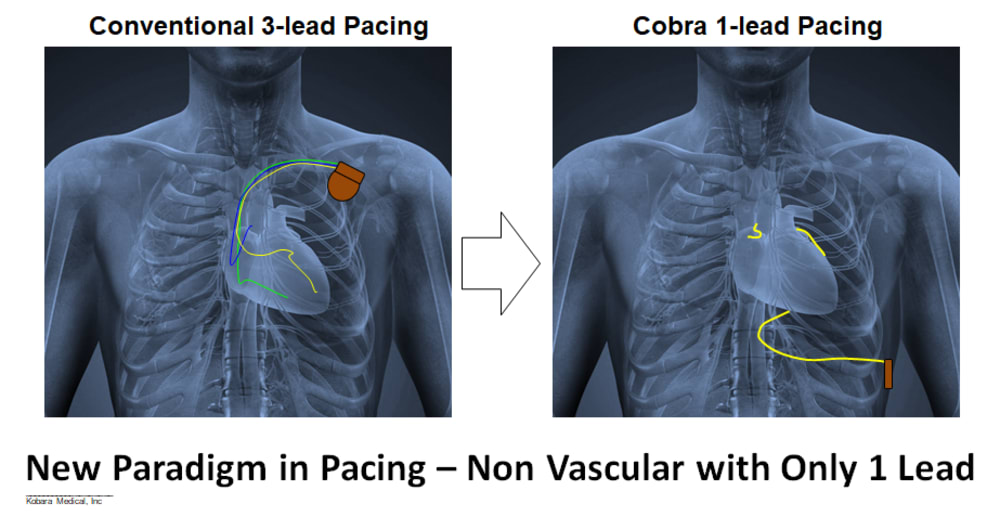
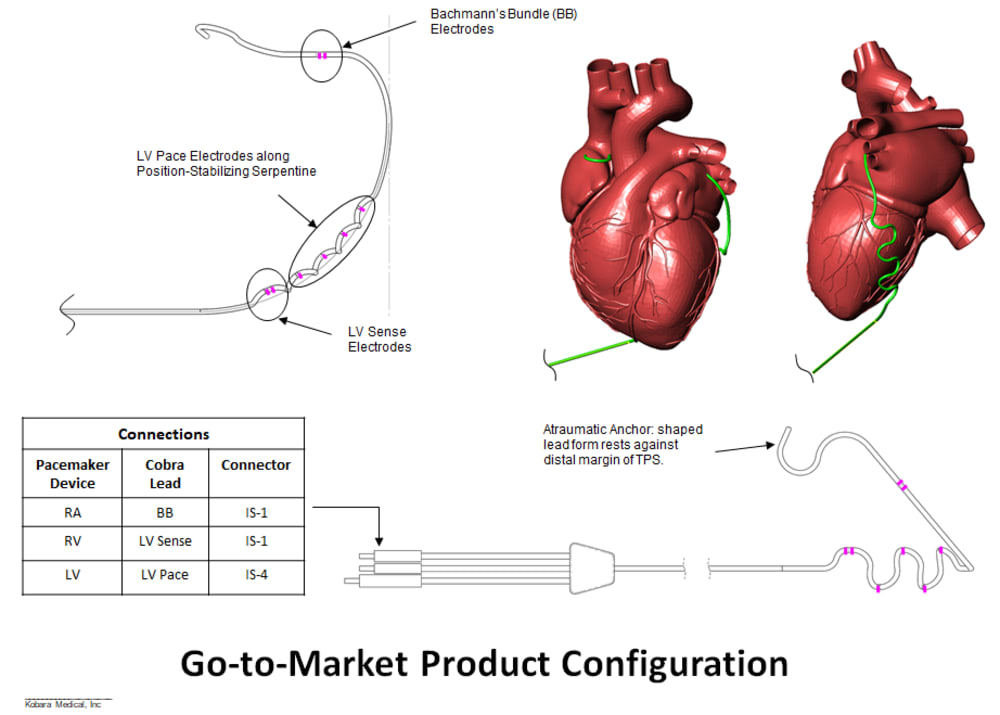
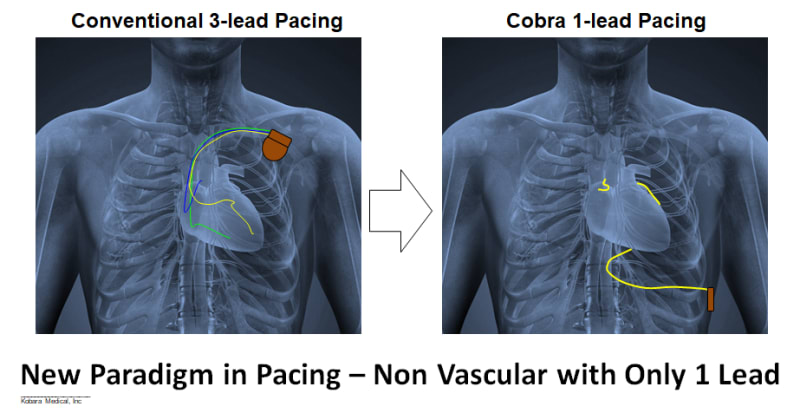
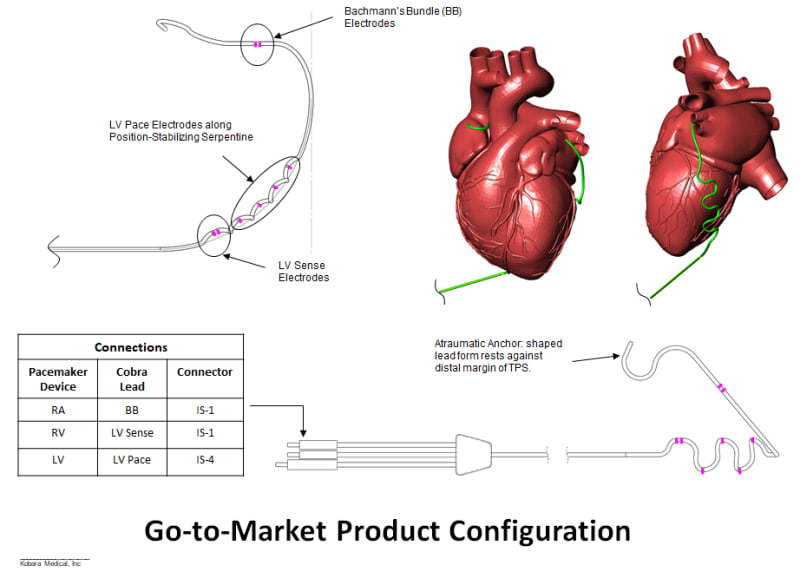
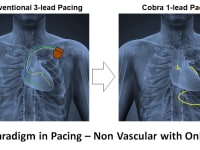
Product Overview: Kobara Medical, Inc is addressing the unmet need by developing an atraumatically anchored multi-chamber and multi-site epicardial lead that makes novel use of the heart’s transverse pericardial sinus (TPS). The first product is configured to plug into existing pacemakers, which provides a very attractive go-to-market strategy. The lead is epicardial so completely outside the heart. Non-coronary placement of the left ventricle (LV) electrodes improves LV pacing with no detrimental right ventricle pacing. Pacing Bachmann's bundle is possible from within the TPS and synchronizes the left and right atria. Finally, the solution is a single lead replacing 3 conventional leads and thereby costs less.
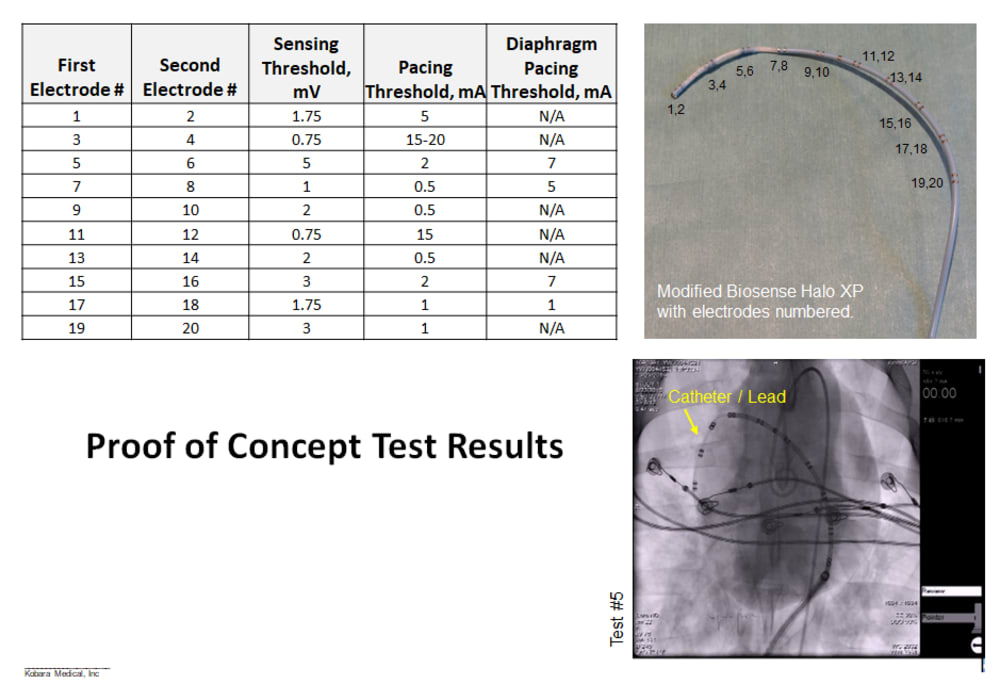
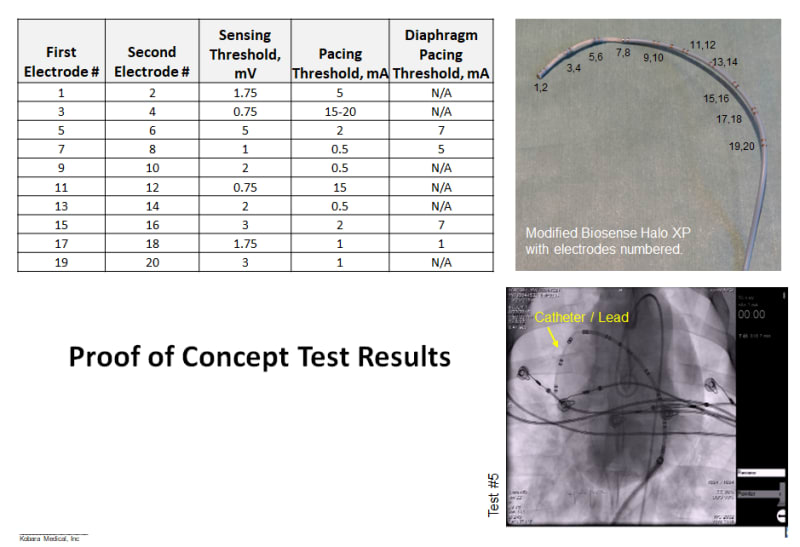
The concept idea has been evaluated in an acute animal study using a decapolar catheter with clinically relevant pacing threshold results, which were presented at the recent Heart Rhythm Society in May 2017. A patent application has been filed.
Business Strategy: Kobara will maintain a virtual business structure using outsourced partners. We will develop the first products as stand-alone leads, since they can work with any existing dual and tri chamber pacemaker devices. Expanded products may include configurations with a shock coil (defibrillation). The regulatory pathway is a PMA and immediate next steps are to develop a fully implantable prototype to verify position and pacing thresholds over a 6 to 8 week time period in an animal. Boston Scientific has agreed to provide their Valitude X4 pacemaker for this study.
Impact: Kobara Medical Inc aims to dramatically reduce complications and improve cardiac rhythm outcomes associated with pacemaker systems, while reducing the overall cost of treatment.
-
Awards
-
 2017 Top 100 Entries
2017 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Andreas Pfahnl
- Type of entry:teamTeam members:Venkatakrishna Tholakanahalli, MD Andreas Pfahnl, ScD John Allen
- Software used for this entry:Solidworks
- Patent status:pending