Problem:
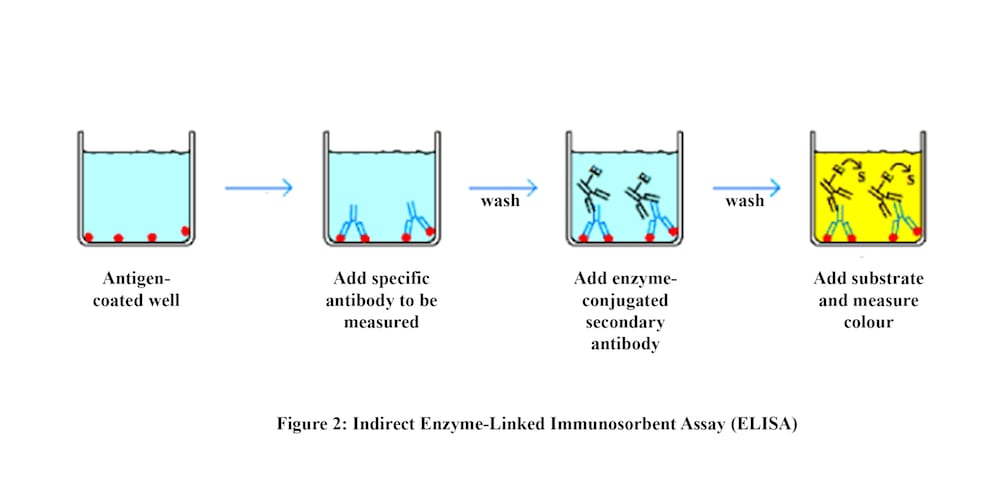
Early detection of diseases increases the chances of treatment significantly. However, regular check-ups can be time-consuming and costly especially in developing countries. This makes Point of Care (PoC) diagnostics very plausible. One commonly used test is ELISA. It incorporates several steps. The blood sample is first introduced into the test well where antigens of the disease are immobilised. If the person is infected, antibodies are present and bind to the immobilised antigens. Second, a buffer solution is used to wash away any unbound antibodies. Third, enzyme-linked antibodies are introduced into the well. They bind to the primary antibodies if present. A washing step is done again. Finally, a substance containing the enzyme's substrate is added. If the person is infected, the substrate changes its colour indicating a positive test. The test is either done manually making it prone to error and dependent on the technician skill or automated requiring costly equipment. A single test costs around 100$. So, a cheap, user-friendly and fully-automated ELISA test is required.
Current Solutions:
Microfluidic-based devices have been used to address the PoC requirements. They use smaller amounts of reagents and have relatively cheap manufacturing cost. However, the current PDMS-based designs are not user-friendly and require an expensive imaging system to detect the signal. Other solutions that are paper-based employ designs for sequential delivery of reagents that reduce the signal and accuracy of the test significantly.
Proposed Solution:
The proposed device is a paper-based microfluidic device. It has five channels each corresponding to one of the reagents in the ELISA process. Fluid flows by capillary action so no pumps are required. All the reagents are loaded initially by the manufacturer. The user only loads the blood sample then presses the start button. It's a single-use button. Two pieces of paper with a void in between form a channel when some mechanical pressure presses them together.
Since ELISA requires sequential delivery of reagents, an absorbent pad is added to each channel. It keeps absorbing the liquid until it is totally full, then flow continues normally. The delay time is controlled by the material and dimensions of the pad.
To cut the channel off after the delivery of the required amount of reagent, a bridge of soluble polymer is used. As the liquid flows, the bridge dissolves in the liquid until it is totally dissolved and the flow stops. All reagents reach the detection spot sequentially and a change in color could be observed by the naked eye in case of infection.
Nitrocellulose is used for the substrate because of its binding capabilities with the antigens. Cellulose is used for the absorbent pads due to its high absorptivity. Mannose is used for the bridge. The channels are defined on the substrate by a hydrophobic material like wax. Wax printing is the technique used for fabrication.
The device costs 3$ and is user-friendly with reliable results. Several detection wells for different diseases could exist on the same test paper enabling detection of different diseases at the same time.
Video
Like this entry?
-
About the Entrant
- Name:Ahmed Elmenshawy
- Type of entry:teamTeam members:Ahmed Abu Bakr Ahmed ElMenshawy Karim ElKholy Mustafa Amer Nader Hozayn
- Software used for this entry:COMSOL Multiphysics
- Patent status:none