Culture patch with monolayer nano-filers on a frame in-vivo like extracellular matrix made of monolayer nanofibers is suited for cell culture and tissue formation. It allows minimizing the exogenous contact and maximizing the uptake rate cells, while the micro-frame and the patch form allow easy handling, easy control of self-organization. Honeycomb frames of different size, shape and elasticity are producible. Nanofibers made of different materials such as gelatin, collagen, PLGA, PCL, PMGI, etc. are also available. The culture patch can be placed in Petri-dish and multi wells for off-ground cell culture. It can also be inserted in Boyden chambers or microfluidic chips. Cells are loaded on the honeycomb frame area and the patch with cells will then be placed in the medium. After culture, the patch can be taken for imaging and other type characterization.
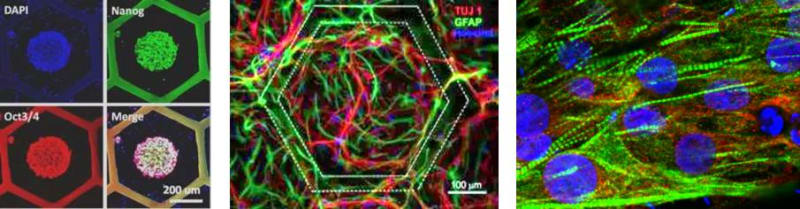
Tissue patches are derived from differentiated human induced pluripotent stem cells (hiPSCs) on nanofibers under off-ground culture conditions, which enabled a more efficient uptake of nutrients and a more efficient cell-organization. Handling and integration of the tissue patch are easy for both in-vitro and in-vivo assays. Both flat and hemisphere hiPSC colonies can be generated in each of the honey compartment, resulting in a more accurate commitment and more homogeneous differentiation. Tissue patches of functional cardiomyocytes are produced, showing up-regulated cardiac gene expression and regular beating over the whole patch area. When placed on a multi-electrode array, characteristic signals and appropriate drug responses could be demonstrated. Similarly, motor neurons are produced on the patch, showing up-regulated expression of neuron specific proteins and spontaneous firing which can be imaged and monitored by extracellular potential recording. General speaking, the metabolic activities of the cells are enhanced on the patch comparing that on a dish, giving rise to a fast increase of the cell number. Co-culturing two types of the hiPSC derived cells, each on one side of the patch, leads to the formation of a double cellular layer that is requested for different tissue models. Furthermore, several tissues patches can be stacked for three-dimensional tissue modeling.
Integration of a tissue patch into a chip gives rise to organ on-a-chip models. Typically, a tissue patch made two types of cells are inserted in the interface of two elastoplastic plates. After clamping, solutions are injected to create physiological conditions. One example is alveolus modeling. By differentiating hiPSCs into epithelial and endothelial cells on upper and lower side of a culture patch, an alveolar tissue is obtained and then integrated into a microfluidic devise to recapitulate the pulmonary physiological conditions. By injecting cell factors and/or nano-materials, it is possible to study the translocation effect at the alveolar epithelium level. More generally, integration of several tissue patches of different cell types into a single microfluidic device will allow creating a body-like circulatory and investigating interaction among different tissues. For example, one can co-culture of human intestine, liver, skin and kidney cells with a medium circulation loop for drug testing.
-
Awards
-
 2018 Top 100 Entries
2018 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Li Wang
- Type of entry:individual
- Patent status:patented