Clinical Motivation: Heart failure, a clinical syndrome resulting in impairment of ventricular filling or the ejection of blood is the leading cause of morbidity and mortality across the world. Left ventricular remodeling, a mechanistic response to myocardial injury, has been shown to underlie the progression to heart failure. As such, sensitive techniques to track the rate and extent of remodeling are necessary to evaluate risk and treatment options on a patient-specific basis. Echocardiography has become the gold standard for assessing the structure and function of the heart. Moreover, recent advancements in both hardware and software have given rise to a relatively new echocardiographic capability: the assessment of regional myocardial deformation through two-dimensional speckle tracking echocardiography. While this study has been shown to be clinically useful in a variety of disease cases, a dependency on hemodynamic load and left ventricular geometry has diminished its widespread clinical utility.
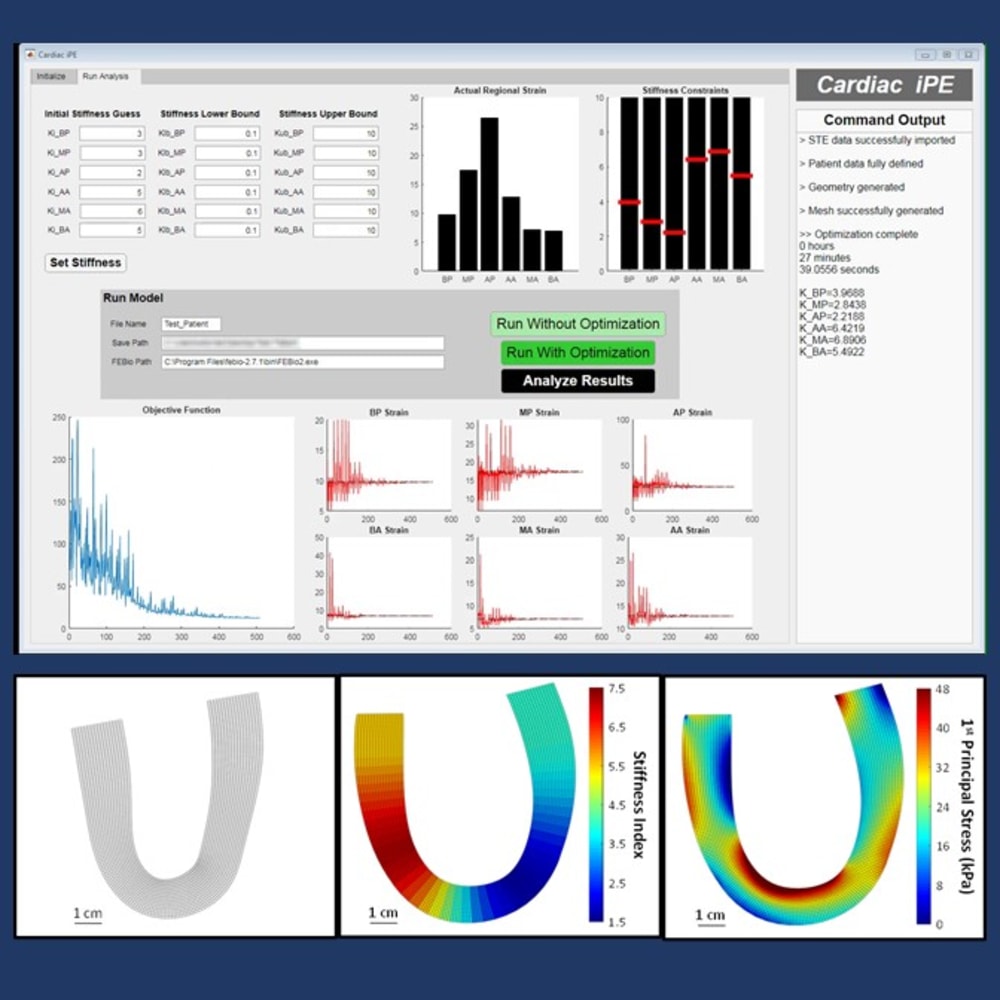
Proposed Technology: The primary motivation behind this novel technology is to non-invasively quantify the local mechanical properties of the left ventricular myocardium, effectively removing the dependency on left ventricular hemodynamics/geometry and potentially providing a more sensitive response variable to track the progression of left ventricular remodeling. An inverse finite element analysis technique is employed with a pattern search optimization algorithm to identify an optimal distribution of mechanical properties necessary to match in-silico strains, average wall thickness, and left ventricular area with those experimentally measured in-vivo. After minimization of this unique objective function, it is possible to then visualize the two-dimensional distribution of mechanical properties across the left ventricle along with a continuous distribution of principal stresses and strains.
Clinical Application: This framework enables the identification of the passive mechanical properties of the intact left ventricular myocardium and therefore could help evaluate the progression of heart disease. This technology will be used as a compliment to standard echocardiographic analysis as a sensitive biomechanical marker of the rate and extent of left ventricular remodeling. Furthermore, this analysis can be completed as a post-processing step from echocardiographic images that are routinely acquired in a complete transthoracic echocardiographic study. This type of point-of-care analysis has the potential to significantly improve cardiovascular disease diagnostics and inform clinicians and researchers on patient-specific disease progression.
Like this entry?
-
About the Entrant
- Name:William Torres
- Type of entry:individual
- Patent status:pending