Digital and mobile technology provides consumers with access to their own personal health data. From measuring the number of steps per day or the number of solid sleep hours per night, consumers have begun to take an active role in tracking their health. Vital sign tracking is critical for monitoring general daily wellness. There is an enormous market opportunity for remote monitoring of chronic and other conditions such as congestive heart failure, COPD, asthma, and diabetes, with the latter estimated to cost the US health care system $582 Billion in 2018. The VitalWellness Platform helps the consumer to do just that. By providing an easy to use monitoring platform, the VitalWellness Platform is perfectly positioned to capture part of this growing market by enabling geriatric and chronically ill patients to monitor their health outside of the hospital and their doctor’s office.
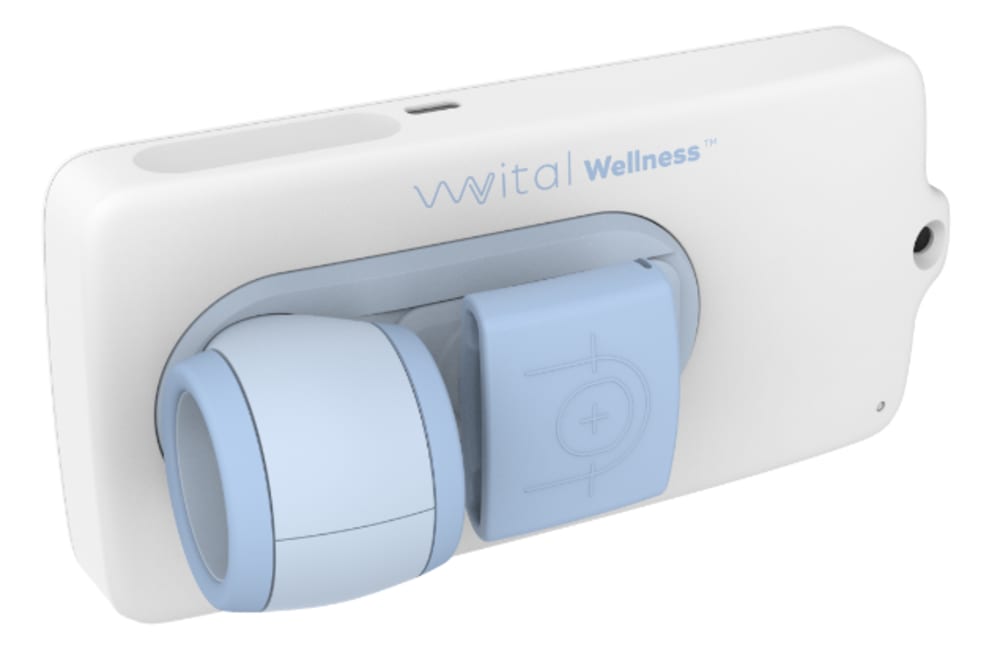
The VitalWellness Platform consists of the VitalWellness device which is an integrated multi-vital sign device which accurately measures blood pressure, pulse rate, SpO2, respiratory rate, heart rate variability, and temperature. The VitalDetect device captures vital signs through a combination of sensors. Temperature is calculated through the 510(k)-cleared InstaTemp MD infrared (K152905) sensor and proprietary body temperature algorithm. Blood pressure is calculated using an inflatable finger cuff (similar method to the blood pressure cuff on the arm) and dynamic light scattering (DLS) sensor subsystem which measure blood flow after occlusion to calculate systolic and diastolic blood pressure. The photoplethysmogram (PPG) subsystem calculates heart rate and SpO2 from the ratio of 660 [nm] and 940 [nm] light absorption in the finger. Respiratory rate is calculated from a combination of the DLS and PPG subsystems. The algorithms for the determination of the vital signs are owned by the Company. The VitalWellness accuracy has been validated for home monitoring use by Partners Connected Healthcare (Boston, MA).
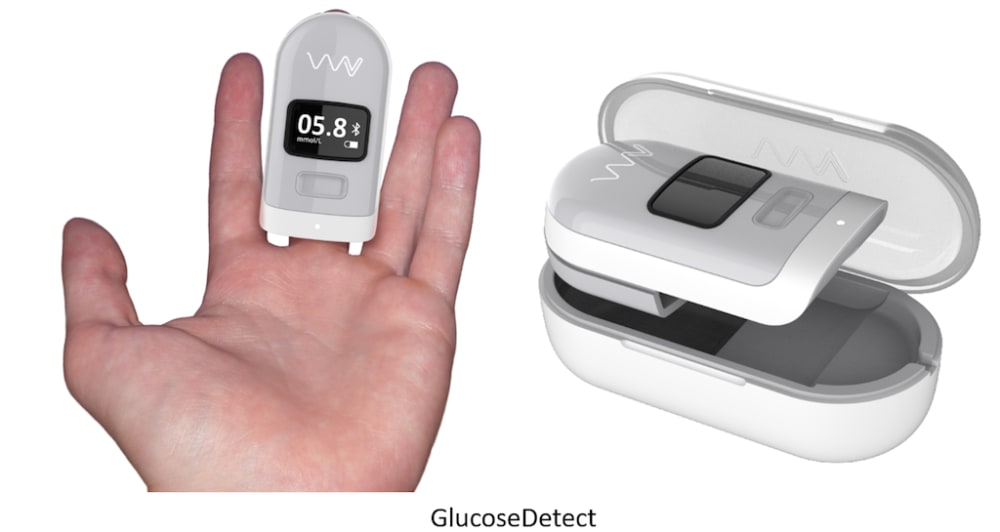
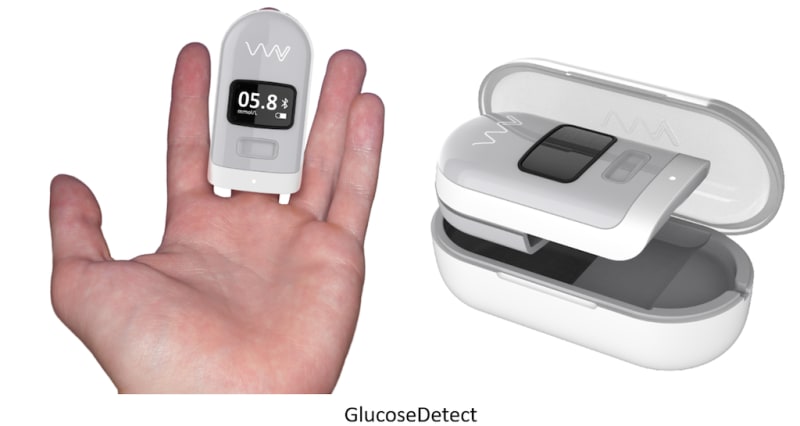
The VitalWellness Platform also consists of the GlucoseDetect device which measures non-invasive glucose through proprietary light scattering emitters and receivers. Monitoring blood glucose levels non-invasively and cost effectively will change the lives of tens-of-millions of people worldwide. This will eliminate the need for finger pricks which will enable more users to take care of their heath and enable painless night-time monitoring of hypoglycemia to prevent “dead in bed” syndrome.
The VitalWelless Platform includes the VitalWeight scale which measures weight and body mass index (BMI) which is crucial for patients with congestive heart failure.
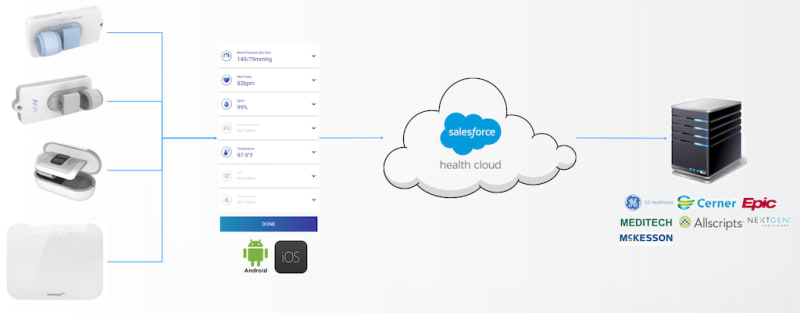
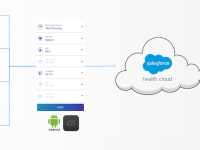
The VitalDetect, GlucoseDetect, and VitalWeight transmit the readings via Bluetooth to the Vital App (Android/iOS) which provides an intuitive guide through the workflow. The results from the Vital App are automatically stored to the Vital Cloud powered by Salesforce Health Cloud and can be reviewed remotely by the patients’ healthcare provider or concerned family member. The Vital App graphically tracks and shows trends of physiological vital signs. In the future, the Vital App will also include a virtual health coach which will be able to provide the patient with the interpretation of the results which will drive behavior.
Video
-
Awards
-
 2019 Top 100 Entries
2019 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Mark Khachaturian, Phd
- Type of entry:teamTeam members:Mark Terrill, CEO (Vital USA)
Brendan Murray, Project Engineer
Edward Hawley, Dir or Quality Assurance
Mary Ann Greenawalt, Dir of Regulatory Affairs - Patent status:patented