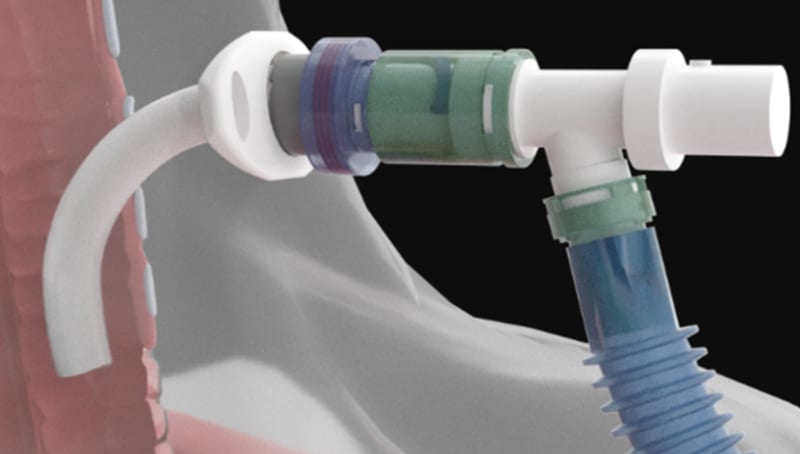
THE PROBLEM: Ventilator-dependent patients frequently experience accidental ventilator disconnect between the tracheostomy tube and the ventilator; a deadly problem known clinically as "pop-off". This accidental “pop-off” can occur multiple times per day, sometimes every hour for some patients. Hospital clinicians have stated that ventilator-dependent patients must stay in the Critical Care Unit (CCU) or the Intensive Care Unit (ICU) to have someone within reach to immediately reconnect the breathing tube to the ventilator when it “pops-off”. Similar scenarios occur for ventilator-dependent patients that are receiving care in a home-care and long-term care environment. If not reconnected within two to four minutes, the patient will die, or suffer catastrophic (permanent) brain injury.
OUR SOLUTION: The POPSafe™ Tracheostomy Tube Set is patented medical device that eliminates these deadly accidental ventilator “pop-off” disconnect events. Our POPSafe™ Tracheostomy Tube Sets are the only products addressing the failure root cause of accidental ventilator disconnections. The POPSafe™ Tracheostomy Tube Sets will be the first and only FDA 510(k) cleared tracheostomy tube sets which include the patented ventilator elbow connector and patented over-pressure relief safety valve. POPSafe™ is a game-changer for ventilator-dependent patients (and their families) because "There are no pop-offs with POPSafe!"
The target audience for our POPSafe™ Tracheostomy Tube Set is the ~250,000 ventilator-dependent patients in the US and their families. The US market size for this product type is $1.5B annually. Of these 250,000 patients, ~63% of these patients will utilize our product in home-care or long-term care facilities; and ~ 37% of these patients receive care in hospitals, including ICUs and CCUs.
POPSafe™ Tracheostomy Tube Sets include several components and are prescription devices. The POPSafe™ Tracheostomy Tube Set will cost the same as current products, and will be priced at ~$170, which is within the Medicare reimbursement price range for current standard of care tracheostomy device sets. The annual spend per patient (ASP) for tracheostomy tube sets is ~$6,000, and is reimbursed by Medicare.
We have developed advanced POPSafe™ prototypes and successfully conducted simulated clinical use testing (mannequins) with clinicians, with compelling results. We have angel investments from an ENT surgeon and RN, who have dealt with their patient’s “pop-off” events, first hand. We’ve been asked to partner with a veteran-owned medical device manufacturer and distribution company. We continue to develop POPSafe™ per FDA design control requirements. The 2019 US Dept. of Veterans Affairs, Office of Inspector General’s Office of Healthcare Inspections report on the investigation of a ventilator-dependent patient death determined “pop-off” to be the primary cause for the patient’s death. One of the five OIG recommendations from this report is to implement policies for use of ventilator anti-disconnect devices; POPSafe is a ventilator anti-disconnect device, which will be GMP manufactured similarly to currently available tracheostomy tube devices.
Video
-
Awards
-
 2021 Medical Honorable Mention
2021 Medical Honorable Mention -
 2021 Top 100 Entries
2021 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Kay Fuller
- Type of entry:individual
- Patent status:patented