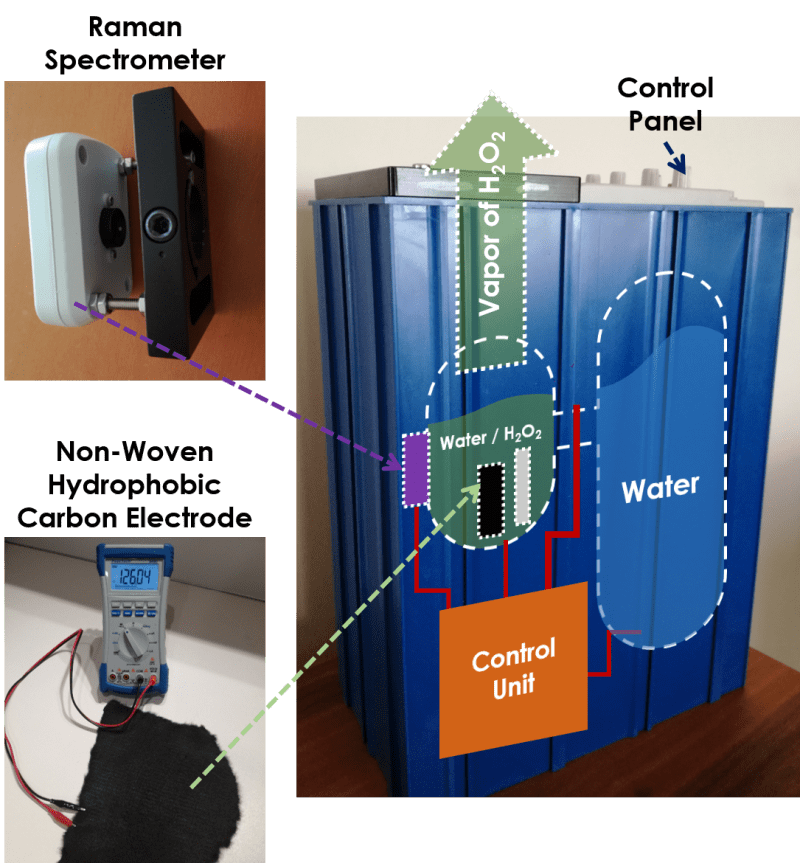
Hydrogen Peroxide (H2O2) is widely used in areal hygiene as in hospitals, schools and other indoor spaces. Also, sanitizing several equipments with H2O2 vapor brings cost efficiency compared to UltraViolet-C (UVC) lamp or LED systems.
We produced a hydrophobic non-woven carbon fiber fabric, decorated with graphene quantum dots. Helmholtz interface composed of hydrophobic surface drives 2-electron reduction that yield higher H2O2 output respect to other catalysts. We obtained roughly 0.34 molal H2O2 in distilled water in 1 minute. This relatively high yield is explained with extended potential window of water induced by hydrophobic layer. Also around 1600m2/g surface area of non-woven hydrophobic Carbon electrode introduce more reduction sites. Though, we are not sure if inner regions are effective in this reaction. In fact, this layer is diminished in further uses unless distilled water is used as source.
Determining H2O2 percentage in water is challenging. One option is to use pH meters, though, maintainance and calibration are challenging and introduce complexity. Also there are H2O2 chemical sensors, but they are costly. We built a low-resolution Raman spectrometer, mounted on transparent reservoir wall. To lower the overall cost and optical complexity, high optical power stimulation is beneficial to discard notch filters in setup. Scattered light is spatially filtered and directed to grating. This, of course, ends up fading the intensity of shift peaks; thus a 1 Watt 405nm LASER is used in pulse-mode to discriminate water (~407nm) and H2O2 (~420nm) shift peaks. Using the ratio of those two peaks, device obtains H2O2 percentage, empirically obtained for different temperatures.
Divulged production rate above indicates around 1 Liter 33% H2O2 water solution output in 28 minutes. Unlike commercial Vaporized Hydrogen Peroxide (VHP) systems, it can safely align concentration in ppm (parts-per-million).
Our product can also find applications in rockets, too. Although we have not investigated, rich H2O2 solutions have been in use in several engines in fuel oxidation.
Like this entry?
-
About the Entrant
- Name:Berker Husam
- Type of entry:individual
- Patent status:none