Corrosion has compromised structural materials since the dawn of civilization. Today, the annual direct cost of metallic corrosion is estimated to be ~$300 billion in the USA alone. Steels are one of the most widely used metals in production and manufacturing, utilities, transportation, defense, and infrastructure; however, they are susceptible to a diverse range of corrosive environments catalyzed by oxygen, moisture, electrolytes, and microbes. It remains a long-standing challenge to develop a universal coating that can protect steel surfaces in both abiotic as well as biotic corrosive environments. For example, while inorganic commercial coatings (e.g., zinc- and chromium-based) and polymer-based coating (e.g., epoxy and polyaniline) form a protective barrier against moisture and Chloride ions, they are ineffective against bacterial environment. Irrespective of the coating material, the presence of ‘Defects’ in any coating will significantly influence corrosion behavior and moderate service life of the coated surface. Therefore, there is an urgent need for an effective corrosion-resistant coating to protect structural materials (both in defect-free and defective condition) from a diverse array of corrosive species, under biotic and abiotic mediums. To address this critical need, we have developed a lightweight low-cost sulfur-selenium (S-Se) alloy which serve as an excellent corrosion-resistant coating with protection efficiency of ~99.9% for steel in a wide range of diverse environments. The coating is strongly adhesive, mechanically robust, and demonstrates excellent damage recovery properties.
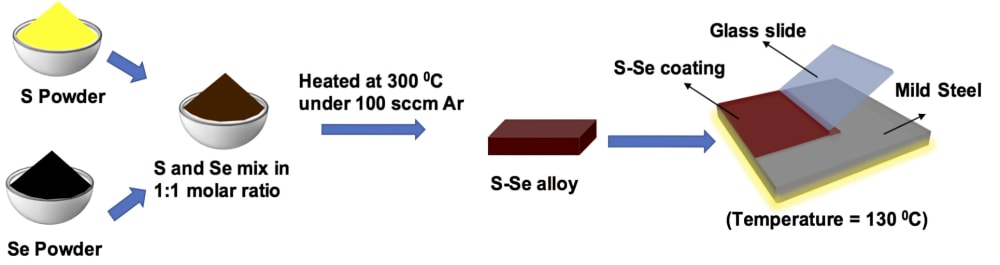
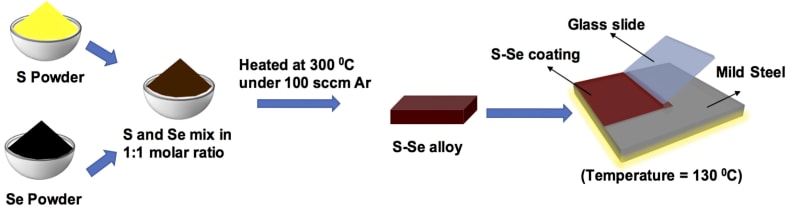
S-Se alloy has been synthesized using elemental sulfur and selenium in a 1:1 molar ratio and then heating to ~300 °C in an inert atmosphere. Initially, the thin coatings on mild steel were applied by using a doctor blade method. It can also be deposited on components with realistic geometries using scalable spray deposition. S-Se coated mild steel showed a corrosion rate that is 6-7 orders of magnitude lower than bare metal in abiotic (simulated seawater and sodium sulfate solution) as well as biotic (sulfate-reducing bacterial medium) environments. The high corrosion resistance of the alloy in diverse environments, stems from its semi-crystalline, non-porous, anti-microbial, dielectric, and viscoelastic nature. Mechanical testing of the alloy reveals markedly high modulus and hardness, far greater than most polymer and nanocomposite coatings, while also maintaining its flexible nature, unlike inorganic coatings. Further, the constituents possess inherent anti-microbial properties and do not dissolve in typical solvents, making it an ideal anti-corrosive coating for disparate environments. Finally, the alloy coating possesses a remarkable ability to repair damage and recover the coating performance. Such phenomena provide the added advantage of significantly reducing the probability of defects being generated and sustained, thus improving its longevity, and corrosion resistance under diverse circumstances.
Corrosion costs equivalent to about 3–4% of each nation's gross domestic product (GDP). Using a 3.4% of global GDP, the global cost of corrosion can then be estimated to be US$2.5 trillion. It is estimated that savings of 15~35% of the cost of corrosion, i.e., ~US $375-$875 billion annually on a global basis could be realized using this all-in-one new coating.
-
Awards
-
 2022 Manufacturing/Robotics/Automation Honorable Mention
2022 Manufacturing/Robotics/Automation Honorable Mention -
 2022 Top 100 Entries
2022 Top 100 Entries
Like this entry?
-
About the Entrant
- Name:Muhammad M Rahman
- Type of entry:teamTeam members:Sandhya Susarla, Md. Abid Shariar Saadi, Nancy Cui, Pulickel M. Ajayan, Muhammad M. Rahman
- Patent status:none